Vertex Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for VERTEX PHARMS, and what generic alternatives to VERTEX PHARMS drugs are available?
VERTEX PHARMS has eight approved drugs.
There are forty-nine US patents protecting VERTEX PHARMS drugs.
There are seven hundred and seventy-nine patent family members on VERTEX PHARMS drugs in fifty-four countries and eighty-four supplementary protection certificates in nineteen countries.
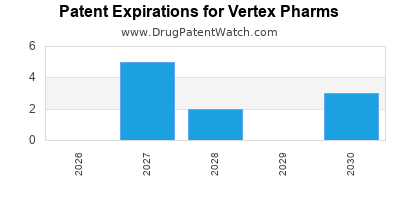
Drugs and US Patents for Vertex Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | ORKAMBI | ivacaftor; lumacaftor | GRANULE;ORAL | 211358-002 | Aug 7, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Vertex Pharms Inc | TRIKAFTA (COPACKAGED) | elexacaftor, ivacaftor, tezacaftor; ivacaftor | TABLET;ORAL | 212273-001 | Oct 21, 2019 | RX | Yes | Yes | 10,239,867 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Vertex Pharms Inc | ORKAMBI | ivacaftor; lumacaftor | GRANULE;ORAL | 211358-001 | Aug 7, 2018 | RX | Yes | No | 7,495,103 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-005 | May 3, 2023 | RX | Yes | No | 8,324,242 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Vertex Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vertex Pharms | INCIVEK | telaprevir | TABLET;ORAL | 201917-001 | May 23, 2011 | 8,529,882 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Premature patent expirations for VERTEX PHARMS
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for Vertex Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Poland | 3219705 | ⤷ Try a Trial |
| Mexico | PA06008606 | ⤷ Try a Trial |
| Uruguay | 37513 | ⤷ Try a Trial |
| Hong Kong | 1152707 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Vertex Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1320540 | 1290007-2 | Sweden | ⤷ Try a Trial | PRODUCT NAME: TELAPREVIR OCH FARMACEUTISKT GODTAGBARA SALTER OCH/ELLER SOLVAT AV TELAPREVIR ELLER SOLVAT AV SADANA SALTER; REG. NO/DATE: EU/1/11/720/001 20110919 |
| 1320540 | 122012000015 | Germany | ⤷ Try a Trial | PRODUCT NAME: TELAPREVIR UND JEDE THERAPEUTISCH AEQUIVALENTE FORM DAVON, WELCHE DURCH DAS GRUNDPATENT GESCHUETZT IST, WIE PHARMAZEUTISCH VERTRAEGLICHE SALZE UND/ODER SOLVATE VON TELAPREVIR ODER SOLVATE SOLCHER SALZE.; REGISTRATION NO/DATE: EU/1/11/720/001 20110919 |
| 3170818 | 2090033-8 | Sweden | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF (A) 3-(6-(1-(2,2-DIFLUOROBENZOD1,3DIOXOL-5-YL)CYCLOPROPANECARBOXAMIDO)-3-METHYLPYRIDIN-2-YL)BENZOIC ACID AND (B) N-(5-HYDROXY-2,4-DITERT-BUTYL-PHENYL)-4-OXO-1H-QUINOLINE-3-CARBOXAMIDE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REG. NO/DATE: EU/1/15/1059 20151124 |
| 2404919 | CR 2016 00019 | Denmark | ⤷ Try a Trial | PRODUCT NAME: 3-(6-((1-(2,2-DIFLUOR-1,3-BENZODIOXOL-5-YL)CYCLOPROPANCATBONYL)AMINO)-3-METHYLPYRIDIN-2-YL)BENZOSYRE, ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, ELLER ET ESTERPRODRUG DERAF; REG. NO/DATE: EU/1/15/1059 20151124 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.