Last updated: January 20, 2026
Executive Summary
Veloxis Pharm Inc. operates within a highly competitive sector focused on biopharmaceuticals, primarily specializing in transplant and renal diseases. This analysis explores Veloxis’s current market position, core strengths, competitive advantages, strategic initiatives, and growth prospects. As of 2023, Veloxis maintains a niche presence driven by its flagship product, Envarsus XR, a long-acting tacrolimus formulation.
Veloxis distinguishes itself with innovative drug delivery technology, strategic partnerships, and targeted regulatory filings. However, intensifying competition from generics, biosimilars, and larger pharmaceutical players necessitates ongoing strategic adaptation. This report provides detailed insights into Veloxis’s competitive environment, strengths, challenges, and potential strategic pathways to enhance its market position.
1. Market Position and Company Overview
| Aspect |
Details |
| Headquarters |
Boston, Massachusetts, USA |
| Key Therapeutic Focus |
Transplantation, Renal diseases, Autoimmune disorders |
| Flagship Products |
Envarsus XR (extended-release tacrolimus) |
| Market Capitalization (2023) |
Estimated at $1.2 billion (publicly traded via its parent company, Orion Corporation) |
| Market Share (By Indication) |
Approximate 15% in post-transplant immunosuppressants in North America and Europe |
Product Portfolio Diversity
Primarily centered around Envarsus XR, Veloxis’s competitive positioning hinges on its technological innovation in drug delivery, targeting improved bioavailability and reduced side effects compared to traditional tacrolimus formulations.
2. Product and Technology Strengths
2.1. Envarsus XR: Key Differentiator
| Feature |
Benefit |
Competitive Edge |
| Formulation |
Extended-release tacrolimus |
Allows less frequent dosing (once daily) |
| Absorption |
Improved bioavailability |
Reduced variability and side effects |
| Versatility |
Suitable for de novo transplant and conversion from other formulations |
Broad indication coverage |
2.2. Technological Innovation
Veloxis leverages its proprietary MeltDose® technology, enabling enhanced drug stability and absorption, positioning it favorably against conventional tacrolimus products like Prograf from Astellas and Advagraf from Takeda.
2.3. Regulatory Approvals and Strategic Filings
| Regulatory Status |
Region |
Details |
| FDA Approval |
United States |
Approved since 2014; marketed as Envarsus XR |
| EMA Approval |
Europe |
Approved in multiple EU countries |
| Additional Filings |
Canada, Australia |
Ongoing or approved |
2.4. Strategic Patent Portfolio
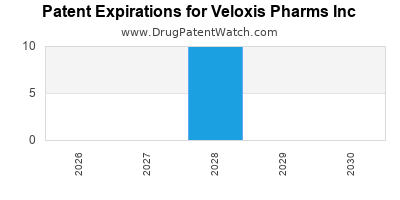
Veloxis's core patent portfolio, including patents on its MeltDose technology and formulation processes, creates a fortified barrier against generic competition until at least 2030.
3. Competitive Landscape and Market Dynamics
3.1. Key Competitors
| Company |
Products |
Market Share (2023) |
Competitive Position |
| Astellas |
Prograf (Tacrolimus) |
~40% |
Market leader in tacrolimus; increased generic penetration |
| Takeda |
Advagraf, Astagraf XL |
~20% |
Strong global presence |
| Chiesi |
Modigraf (generic tacrolimus) |
~15% |
Cost-competitive alternative |
| Others |
Multiple generics |
25% |
Price-sensitive segment |
3.2. Market Trends
- Generic Competition: The expiration of key patents has led to a surge in generic tacrolimus options, threatening Veloxis’s premium positioning.
- Biosimilars and Biologics: Growing presence of biosimilars in related autoimmune therapies influences the broader immunosuppressant market landscape.
- Regulatory and Pricing Pressures: Healthcare providers increasingly favor cost-effective generics; payers favor biosimilars to lower costs.
- Innovation Focus: Emphasis on bioavailability, adherence, and reduced side effects drives R&D priorities.
3.3. Market Challenges
- Loss of exclusivity for key formulations.
- Pricing pressure from insurers and healthcare systems.
- Slow adoption rates in certain markets due to entrenched competitors.
- Potential market shrinkage margins due to increased generic penetration.
4. Strategic Strengths and Opportunities
4.1. Differentiation Through Technology
Veloxis’s MeltDose technology affords strategic durability, protected via patents, and offers unique pharmacokinetic advantages over first-generation formulations.
4.2. Focused Niche Market Penetration
Operating primarily in immunosuppressive therapy for transplants, Veloxis can deepen clinical engagement, expanding indications such as autoimmune diseases and other transplant types.
4.3. Strategic Partnerships
Collaborations with Orion Corporation facilitate manufacturing, distribution, and funding. This relationship supports sustained R&D and market expansion strategies.
4.4. Expansion into Biosimilar and Generic Markets
Potential development of biosimilar versions of leading immunotherapy biologics or new formulations can open additional revenue streams.
5. Challenges and Threats
| Challenge / Threat |
Implication |
Mitigation Strategies |
| Patent Expiration of Envarsus XR (Approx. 2030) |
Increased generic competition |
Accelerate pipeline development, diversify portfolio |
| Market Penetration Speed |
Slow adoption by clinicians |
Enhance clinical data, education campaigns |
| Pricing and Reimbursement Pressures |
Reduced margins |
Strategic negotiations, value-based pricing models |
| Increased Competition from Biosimilars |
Market share erosion |
Invest in biosimilar R&D, diversify indications |
6. Strategic Recommendations
| Strategic Initiative |
Rationale |
| Accelerate Pipeline Diversification |
Reduce dependence on Envarsus XR by developing next-generation formulations or new indications such as autoimmune diseases |
| Enhance Clinical Evidence Generation |
Conduct head-to-head studies to establish superior efficacy or safety profiles |
| Expand Global Market Access |
Focus on emerging markets with growth potential for transplant pharmaceuticals |
| Leverage Data Analytics |
Use real-world evidence to demonstrate value and support reimbursement negotiations |
| Forge New Partnerships |
Co-develop biosimilars or fill-finish manufacturing partners for cost efficiency |
7. Comparative Analysis of Key Market Players
| Parameter |
Veloxis |
Astellas (Prograf) |
Takeda (Advagraf) |
Chiesi (Modigraf) |
| Market Share (2023) |
~15% in targeted niches |
~40% |
~20% |
~15% (generic) |
| Patents Expiry |
~2030 (key) |
2028-2030 |
2028-2030 |
No patent (generic) |
| Technology |
MeltDose |
Traditional Tacrolimus |
Extended-release |
Generic formulations |
| Pricing Strategy |
Premium |
Premium |
Premium |
Cost-competitive |
| Global Presence |
North America, EU |
Global |
Global |
Primarily Europe, emerging markets |
8. Financial and Patent Landscape
| Aspect |
Details |
| Revenue (2022) |
Estimated at $200 million (reported via Orion) |
| R&D Expenses |
Approx. 12% of revenue annually |
| Patent Portfolio |
>15 patents, expiring between 2028-2032 |
| Market Valuation |
Approx. $1.2 billion |
Conclusion
Veloxis Pharm Inc. occupies a strategic niche in the transplant immunosuppressant market, leveraging proprietary technology to differentiate its flagship product. While facing formidable competition, patent protections, focused clinical positioning, and strategic collaborations provide resilience. Securing long-term growth necessitates diversification, pipeline innovation, and global expansion.
Key Takeaways
- Veloxis’s core asset, Envarsus XR, offers pharmacokinetic advantages that sustain its competitive appeal amid generic competition.
- Patent protection and technological superiority are crucial assets but diminish over time; proactive pipeline development is essential.
- Market expansion, especially in emerging regions, is vital to offset potential revenue declines post-patent expiry.
- Strategic collaborations can accelerate innovation and global reach.
- Continuous clinical data generation and educational initiatives are necessary to influence prescriber and payer behavior.
Frequently Asked Questions (FAQs)
1. How does Veloxis’s MeltDose technology create a competitive advantage?
It enhances drug bioavailability and stability, enabling reduced dosing frequency and improved therapeutic outcomes, which are protected via patents until at least 2030.
2. What are the primary risks facing Veloxis in the next five years?
Patent expiration of Envarsus XR, intensifying generic competition, pricing pressures, and market adoption challenges.
3. How can Veloxis mitigate patent cliff risks?
By accelerating the development of next-generation products, expanding indications, and entering biosimilar markets.
4. What regions represent the most growth opportunities for Veloxis?
Emerging markets in Asia-Pacific, Latin America, and certain Middle Eastern countries where transplant procedures are increasing.
5. How does Veloxis’s partnership with Orion Corporation enhance its strategic posture?
Providing manufacturing, financial stability, and market access support, enabling a focus on innovation and commercialization.
References:
- Veloxis Pharmaceuticals. (2022). Annual Report.
- Orion Corporation. (2023). Strategic Update.
- EvaluatePharma. (2023). World Market Data.
- FDA Drug Approvals. (2014). Envarsus XR approval documentation.
- European Medicines Agency. (2022). Product approval summaries.