Last updated: February 20, 2026
What is Pharmacyclics LLC’s current market position?
Pharmacyclics LLC operates as a biotechnology company primarily focused on developing and commercializing therapies for hematologic cancers and certain solid tumors. Acquired by AbbVie in 2015 for approximately $21 billion, the company's flagship product is Imbruvica (ibrutinib), a BTK inhibitor used for multiple indications including chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), and other hematological malignancies.
Pharmacyclics primarily leverages Imbruvica's profile to maintain a leading share in the targeted therapies segmentation. As of 2022, Imbruvica generated revenues of $5 billion globally, with a market share exceeding 60% in certain hematology indications.
How does Pharmacyclics compare to competitors?
| Company |
Key Products |
Major Indications |
2022 Revenue (USD) |
Market Share (Primary Indication) |
Focus Area |
| AbbVie (Pharmacyclics) |
Imbruvica (ibrutinib) |
CLL, MCL, Waldenström's macroglobulinemia |
$5 billion |
60% (CLL) |
BTK inhibitors, Hematologic malignancies |
| Johnson & Johnson |
Darzalex (daratumumab) |
Multiple myeloma |
$4.1 billion |
Leading |
Monoclonal antibodies, Hematology |
| Novartis |
Kymriah (tisagenlecleucel) |
ALL, lymphoma |
$800 million |
Niche |
CAR-T therapies |
| Gilead Sciences |
Yescarta (axicabtagene ciloleucel) |
Non-Hodgkin lymphoma |
$720 million |
Niche |
CAR-T therapies |
Pharmacyclics’ Imbruvica rivals include Gilead’s Yescarta and Novartis’s Kymriah, with the latter focusing on cell therapy. In the BTK inhibitor space, AstraZeneca's Calquence (acalabrutinib) presents a key competitor with increasing market share.
What are the core strengths of Pharmacyclics LLC?
- Market Leadership with Imbruvica: The drug has a first-mover advantage in BTK inhibitors for hematological cancers, with a broad label expansion and multiple indications approved globally.
- Strong R&D Portfolio: Despite being acquired by AbbVie, ongoing clinical trials aim to expand Imbruvica’s use into multiple myeloma, CLL, and other lymphoid malignancies.
- Strategic Acquisitions and Partnerships: The 2015 acquisition by AbbVie provided funding and global reach, enabling extensive marketing and ongoing pipeline development.
- Robust Revenue Generation: Imbruvica’s sales consistently surpass other targeted therapies, offering a reliable revenue stream.
- Regulatory Approvals: Imbruvica has received FDA approvals for 10+ indications, facilitating market penetration and clinical adoption.
What strategic insights can be drawn for Pharmacyclics LLC?
- Pipeline Expansion: Focus on differentiating Imbruvica through combination therapies and additional indications, especially in multiple myeloma.
- Competitor Navigation: Keep pace with AstraZeneca’s Calquence in the BTK inhibitor market; consider co-developments or strategic collaborations.
- Global Market Access: Enhance presence in emerging markets experiencing rising hematologic cancer cases, such as China and India.
- Innovation in Adjacent Therapies: Invest in next-generation BTK inhibitors and other kinase inhibitors with improved safety profiles.
- Leverage Data for Precision Medicine: Use real-world evidence and biomarkers to optimize patient selection, ensuring maximal efficacy.
What are the potential threats facing Pharmacyclics LLC?
- Market Saturation: Increasing competition from second-generation BTK inhibitors offering better safety and efficacy, such as AstraZeneca’s Calquence and BeiGene’s Brukvec.
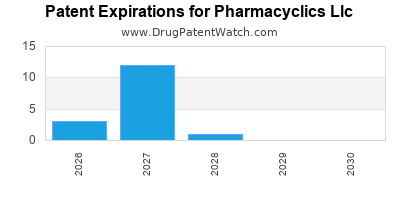
- Patent Expiry Risks: Imbruvica’s patents are nearing expiration in key markets, opening opportunities for biosimilars and generics.
- Regulatory Challenges: Future approvals may face hurdles if safety or efficacy data are challenged.
- Emerging Therapies: CAR-T and other immunotherapies developing rapidly could diminish Imbruvica’s dominance if they demonstrate superior outcomes or reduced adverse events.
- Pricing Pressure: Payers may impose cost constraints, especially as new competitors enter the market.
How does the regulatory environment impact Pharmacyclics LLC?
Imbruvica holds FDA approval for more than 10 indications, with recent approvals extending into new hematological malignancies. The European Medicines Agency (EMA) also approved Imbruvica for similar indications.
Future regulatory pathways focus on accelerated approvals for combination regimens and new indications, contingent on ongoing clinical trial results. Biosimilar competition could influence pricing and reimbursement strategies, especially in Europe and the U.S.
How should Pharmacyclics LLC position itself moving forward?
- Continue clinical development to deepen Imbruvica’s label, particularly targeting multiple myeloma and chronic lymphocytic leukemia.
- Form strategic collaborations to combine Imbruvica with other targeted or immunotherapy agents.
- Expand global access in Asia, Latin America, and Africa to bolster revenue streams.
- Invest in next-generation BTK inhibitors with improved safety profiles to preempt emerging competitors.
- Monitor patent estates closely, prepare for biosimilar entries to mitigate revenue losses.
Key Takeaways
- Pharmacyclics LLC is positioned as a leader in BTK inhibitor therapy, primarily driven by Imbruvica.
- Competition from second-generation agents and immunotherapies represents a significant threat.
- Continued product pipeline development, geographic expansion, and innovation in combination therapies are essential.
- Patent expiry and biosimilar entry will demand strategic pricing and market access initiatives.
FAQs
1. What is the main product of Pharmacyclics LLC?
Imbruvica (ibrutinib), a BTK inhibitor approved for multiple hematologic malignancies.
2. Who are the primary competitors?
AstraZeneca's Calquence, Gilead's Yescarta, and Novartis's Kymriah.
3. What are the growth prospects for Imbruvica?
Expansion into additional indications and global markets; however, patent expiries pose risks.
4. How does Pharmacyclics’ strategy adapt to emerging therapies?
By investing in clinical trials for combination regimens and next-generation inhibitors, plus geographic expansion.
5. What regulatory trends should be monitored?
Approvals for new indications, accelerated pathways, and biosimilar regulations affecting patent protections.
References
[1] IMS Health. (2022). Hematology & Oncology Market Analysis.
[2] AbbVie. (2022). Imbruvica: Product dossier.
[3] FDA. (2022). Drug Approvals and Indications Database.
[4] AstraZeneca. (2022). Calquence clinical trial data.
[5] Novartis. (2022). Kymriah regulatory filings.