Neos Theraps Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for NEOS THERAPS INC
NEOS THERAPS INC has three approved drugs.
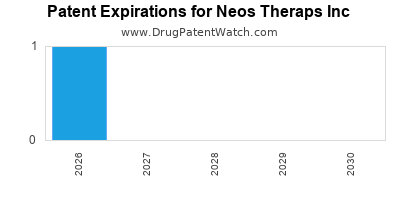
There are seven US patents protecting NEOS THERAPS INC drugs.
There are eight patent family members on NEOS THERAPS INC drugs in five countries.
Drugs and US Patents for Neos Theraps Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neos Theraps Inc | COTEMPLA XR-ODT | methylphenidate | TABLET, ORALLY DISINTEGRATING, EXTENDED RELEASE;ORAL | 205489-004 | Aug 19, 2024 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Neos Theraps Inc | COTEMPLA XR-ODT | methylphenidate | TABLET, ORALLY DISINTEGRATING, EXTENDED RELEASE;ORAL | 205489-001 | Jun 19, 2017 | RX | Yes | No | 8,840,924 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Neos Theraps Inc | ADZENYS ER | amphetamine | SUSPENSION, EXTENDED RELEASE;ORAL | 204325-001 | Sep 15, 2017 | DISCN | Yes | No | 8,709,491 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Neos Theraps Inc | COTEMPLA XR-ODT | methylphenidate | TABLET, ORALLY DISINTEGRATING, EXTENDED RELEASE;ORAL | 205489-003 | Jun 19, 2017 | RX | Yes | Yes | 11,166,947 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Neos Theraps Inc | COTEMPLA XR-ODT | methylphenidate | TABLET, ORALLY DISINTEGRATING, EXTENDED RELEASE;ORAL | 205489-002 | Jun 19, 2017 | RX | Yes | No | 11,166,947 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Neos Theraps Inc | COTEMPLA XR-ODT | methylphenidate | TABLET, ORALLY DISINTEGRATING, EXTENDED RELEASE;ORAL | 205489-002 | Jun 19, 2017 | RX | Yes | No | 9,089,496 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Neos Theraps Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2018085256 | ⤷ Start Trial |
| Australia | 2017353921 | ⤷ Start Trial |
| European Patent Office | 2726066 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2013003622 | ⤷ Start Trial |
| South Korea | 20190107655 | ⤷ Start Trial |
| Japan | 2020504763 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Similar Applicant Names
Here is a list of applicants with similar names.