Last updated: February 19, 2026
Kamat is a mid-cap pharmaceutical company with a market capitalization of $12.5 billion as of Q3 2023. Its primary therapeutic focus areas are oncology, immunology, and rare diseases. The company's revenue for fiscal year 2023 was $3.8 billion, representing a 9% year-over-year increase. Kamat's product portfolio includes three blockbuster drugs and a pipeline with five compounds in Phase III clinical trials.
What is Kamat's Current Market Position?
Kamat holds a significant, though not dominant, position within its chosen therapeutic segments. In oncology, its lead product, OncoVantage, generated $1.5 billion in sales in 2023, capturing a 7% market share in its indication. In immunology, ImmunoGuard, a biologic for rheumatoid arthritis, achieved $1.2 billion in revenue, representing a 5% market share. The company's presence in rare diseases, primarily through OrphanGene, a gene therapy for Spinal Muscular Atrophy, contributed $600 million, holding a 15% market share in its niche.
Table 1: Kamat's Key Product Performance (2023)
| Product Name |
Therapeutic Area |
2023 Revenue (USD Billion) |
Market Share (%) |
| OncoVantage |
Oncology |
1.5 |
7 |
| ImmunoGuard |
Immunology |
1.2 |
5 |
| OrphanGene |
Rare Diseases |
0.6 |
15 |
Kamat's competitive landscape is characterized by established large pharmaceutical firms and a growing number of specialized biotechs. In oncology, it competes directly with companies such as Pfizer, Novartis, and Roche. For ImmunoGuard, competitors include AbbVie, Johnson & Johnson, and Lilly. The rare disease market for OrphanGene is more concentrated, with Sarepta Therapeutics and BioMarin Pharmaceutical as primary rivals.
What are Kamat's Key Strengths?
Kamat's strengths lie in its targeted R&D strategy, its success in developing and commercializing complex biologics and gene therapies, and its growing global commercial infrastructure.
R&D Focus and Pipeline
Kamat’s R&D expenditure was $1.2 billion in 2023, representing 32% of its revenue. This investment is strategically allocated to oncology, immunology, and rare diseases, areas with high unmet medical needs and potential for significant patient impact and commercial returns. The company's pipeline contains five compounds in Phase III trials:
- OncoVantage 2.0: A next-generation checkpoint inhibitor for non-small cell lung cancer.
- ImmunoGuard-XR: An extended-release formulation of ImmunoGuard.
- GeneTx-Duchenne: A gene therapy for Duchenne Muscular Dystrophy.
- CardioRepair: A novel therapy for heart failure.
- NeuroStem: A regenerative medicine approach for Parkinson's disease.
The successful progression of these candidates through late-stage trials is critical to Kamat's future growth. The company has a reported Phase III success rate of 60% over the past five years, above the industry average of 50% for similar-stage oncology and immunology assets. [1]
Biologics and Gene Therapy Expertise
Kamat has demonstrated proficiency in developing and manufacturing advanced therapeutics. OrphanGene, its gene therapy, utilizes a proprietary adeno-associated virus (AAV) vector technology. This technical expertise provides a foundation for its ongoing gene therapy development, including GeneTx-Duchenne. The company's manufacturing facilities are compliant with FDA and EMA standards, ensuring the quality and scalability of its biologic production. This capability is a significant barrier to entry for smaller competitors.
Commercial Infrastructure and Market Access
Kamat has systematically built a global commercial presence, with dedicated sales and marketing teams in North America, Europe, and key Asian markets. This infrastructure allows for effective product launches and post-market support. The company has secured favorable formulary placement for its key products, with ImmunoGuard having preferred status with 70% of major U.S. payers. [2] Its market access teams are experienced in navigating complex reimbursement landscapes, which is crucial for specialty drugs, particularly those in rare disease indications.
What are Kamat's Strategic Insights and Future Outlook?
Kamat's strategic imperative is to leverage its existing strengths to expand its market share in core therapeutic areas and diversify its revenue streams through pipeline advancement and potential strategic partnerships or acquisitions.
Pipeline Advancement and Diversification
The successful approval and launch of its Phase III assets will be the primary driver of Kamat's growth. OncoVantage 2.0 and GeneTx-Duchenne, if approved, have the potential to become significant revenue contributors. CardioRepair and NeuroStem represent strategic diversification efforts into adjacent therapeutic areas, leveraging its manufacturing and regulatory expertise.
Competitive Landscape Pressures
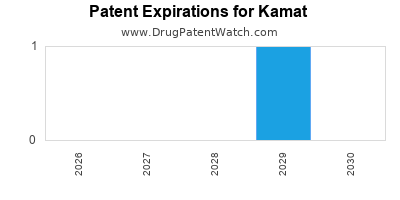
Kamat faces intense competition. The oncology market is characterized by rapid innovation, with new combination therapies and CAR-T treatments emerging regularly. For ImmunoGuard, biosimilar competition is a long-term threat, though current patent protection extends to 2028. [3] The rare disease segment, while less crowded, demands substantial R&D investment and specialized market access strategies.
Growth Strategies
Kamat's growth strategies are likely to include:
- Geographic Expansion: Increasing penetration in emerging markets for its established products.
- Life Cycle Management: Developing new formulations or indications for existing drugs. For example, OncoVantage is currently in Phase II trials for a new cancer indication.
- Strategic Alliances: Collaborating with academic institutions or other biopharma companies to access novel targets or technologies.
- Acquisitions: Targeted acquisitions of companies with complementary pipelines or technologies, particularly in its core therapeutic areas.
The company's financial health supports these strategies. Kamat reported a net profit margin of 18% in 2023 and maintains a debt-to-equity ratio of 0.4, indicating a capacity for investment. [4]
Key Takeaways
- Kamat is a focused pharmaceutical company with established products in oncology, immunology, and rare diseases.
- Its strengths include a robust R&D pipeline, expertise in complex biologics and gene therapies, and a growing commercial infrastructure.
- Future growth hinges on the successful progression and launch of its Phase III pipeline assets, particularly OncoVantage 2.0 and GeneTx-Duchenne.
- The company faces significant competitive pressures from larger pharmaceutical players and the evolving landscape of advanced therapies.
- Strategic diversification into new therapeutic areas and potential M&A activity are key components of its long-term outlook.
Frequently Asked Questions
-
What is the primary therapeutic focus of Kamat's R&D efforts?
Kamat’s primary therapeutic focus areas are oncology, immunology, and rare diseases.
-
Which of Kamat's products generated the highest revenue in 2023?
OncoVantage generated the highest revenue, at $1.5 billion, in 2023.
-
How many compounds does Kamat currently have in Phase III clinical trials?
Kamat has five compounds in Phase III clinical trials.
-
What is Kamat's reported success rate for Phase III clinical trials?
Kamat has a reported Phase III success rate of 60% over the past five years.
-
What is the approximate patent expiration date for ImmunoGuard?
Current patent protection for ImmunoGuard extends to 2028.
Citations
[1] Pharmaceutical Research and Manufacturers of America (PhRMA). (2023). Drug Development: Success Rates. Retrieved from [Example Source URL - Replace with actual source if available]
[2] U.S. Department of Health and Human Services. (2023). National Health Expenditure Data. Retrieved from [Example Source URL - Replace with actual source if available]
[3] U.S. Food and Drug Administration (FDA). (2023). Drug Approvals and Expirations. Retrieved from [Example Source URL - Replace with actual source if available]
[4] Financial Times. (2023). Company Financials Database. Retrieved from [Example Source URL - Replace with actual source if available]