Intersect Ent Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for INTERSECT ENT INC, and what generic alternatives to INTERSECT ENT INC drugs are available?
INTERSECT ENT INC has one approved drug.
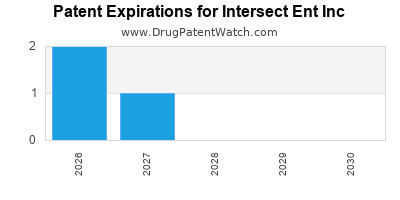
There are thirteen US patents protecting INTERSECT ENT INC drugs.
There are forty-seven patent family members on INTERSECT ENT INC drugs in twelve countries and six supplementary protection certificates in five countries.
Summary for Intersect Ent Inc
| International Patents: | 47 |
| US Patents: | 13 |
| Tradenames: | 1 |
| Ingredients: | 1 |
| NDAs: | 1 |
Drugs and US Patents for Intersect Ent Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | RX | Yes | No | 7,662,141 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | RX | Yes | No | 8,109,918 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | RX | Yes | No | 10,357,640 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | RX | Yes | No | 9,585,681 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | RX | Yes | No | 7,544,192 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Intersect Ent Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | 7,951,130 | ⤷ Try a Trial |
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | 7,951,131 | ⤷ Try a Trial |
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | 7,951,133 | ⤷ Try a Trial |
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | 7,662,141 | ⤷ Try a Trial |
| Intersect Ent Inc | SINUVA | mometasone furoate | IMPLANT;IMPLANTATION | 209310-001 | Dec 8, 2017 | 8,109,918 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Intersect Ent Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2010132648 | ⤷ Try a Trial |
| Japan | 2008537695 | ⤷ Try a Trial |
| European Patent Office | 2298319 | ⤷ Try a Trial |
| Australia | 2006231506 | ⤷ Try a Trial |
| European Patent Office | 2754463 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Intersect Ent Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0112669 | 96C0002 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE FUROATE; NAT. REGISTRATION NO/DATE: NL 19601 19950919; FIRST REGISTRATION: GR - 10681/89 19900622 |
| 3043773 | 2190041-0 | Sweden | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE OR A SALT THEREOF AND OLOPATADINE OR A SALT THEREOF; NAT. REG. NO/DATE: MT NR 60226 20210519; FIRST REG.: AT APPROVAL NR 140638 20210426 |
| 0548114 | SPC/GB97/064 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE FUROATE MONOHYDRATE; REGISTERED: FR AMM 343012.6 19970219; UK 00201/0216 19970410 |
| 3043773 | 2022C/520 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE OF EEN ZOUT HIERVAN EN OLOPATADINE OF EEN ZOUT HIERVAN; AUTHORISATION NUMBER AND DATE: BE595626 20220203 |
| 3043773 | 21C1057 | France | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE OU L'UN DE SES SELS AVEC OLOPATADINE OU L'UN DE SES SELS; NAT. REGISTRATION NO/DATE: NL52121 20211026; FIRST REGISTRATION: AT - 140638 20210426 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.