Hope Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for HOPE PHARMS, and when can generic versions of HOPE PHARMS drugs launch?
HOPE PHARMS has three approved drugs.
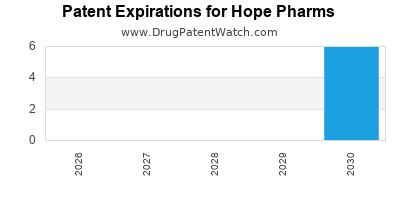
There are seven US patents protecting HOPE PHARMS drugs.
There are forty-one patent family members on HOPE PHARMS drugs in fifteen countries and sixty-seven supplementary protection certificates in eight countries.
Drugs and US Patents for Hope Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hope Pharms | SODIUM THIOSULFATE | sodium thiosulfate | SOLUTION;INTRAVENOUS | 203923-001 | Feb 14, 2012 | RX | Yes | Yes | 8,496,973 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Hope Pharms | NITHIODOTE | sodium nitrite; sodium thiosulfate | SOLUTION, SOLUTION;INTRAVENOUS, INTRAVENOUS | 201444-001 | Jan 14, 2011 | RX | Yes | Yes | 9,585,912 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Hope Pharms | SODIUM THIOSULFATE | sodium thiosulfate | SOLUTION;INTRAVENOUS | 203923-001 | Feb 14, 2012 | RX | Yes | Yes | 10,479,686 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Hope Pharms | NITHIODOTE | sodium nitrite; sodium thiosulfate | SOLUTION, SOLUTION;INTRAVENOUS, INTRAVENOUS | 201444-001 | Jan 14, 2011 | RX | Yes | Yes | 11,753,301 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Hope Pharms | NITHIODOTE | sodium nitrite; sodium thiosulfate | SOLUTION, SOLUTION;INTRAVENOUS, INTRAVENOUS | 201444-001 | Jan 14, 2011 | RX | Yes | Yes | 9,687,506 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Hope Pharms | SODIUM THIOSULFATE | sodium thiosulfate | SOLUTION;INTRAVENOUS | 203923-001 | Feb 14, 2012 | RX | Yes | Yes | 11,753,301 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Hope Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Lithuania | 3569237 | ⤷ Try a Trial |
| Croatia | P20211077 | ⤷ Try a Trial |
| Norway | 2451435 | ⤷ Try a Trial |
| Japan | 6438071 | ⤷ Try a Trial |
| Hungary | E055250 | ⤷ Try a Trial |
| European Patent Office | 2451435 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Hope Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2563920 | CR 2019 00001 | Denmark | ⤷ Try a Trial | PRODUCT NAME: INOTERSEN AND SALT THEREOF, INCLUDING SODIUM SALTS; REG. NO/DATE: EU/1/18/1296 20180710 |
| 0957929 | SPC/GB06/021 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: PEGAPTANIB, PREFERABLY IN THE FORM OF ITS SODIUM SALT; REGISTERED: UK EU/1/05/325/001 20060201 |
| 1874117 | 2014/032 | Ireland | ⤷ Try a Trial | PRODUCT NAME: DOLUTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF, INCLUDING DOLUTEGRAVIR SODIUM; REGISTRATION NO/DATE: EU/1/13/892/001-002 20140121 |
| 2203431 | 15C0013 | France | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OU UN SEL DE CELUI-CI, NOTAMMENT LE SEL DE SODIUM; REGISTRATION NO/DATE: EU/1/14/983 20150119 |
| 0290047 | 97C0108 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MANGAFODIPIR TRISODIUM (ANHYDROUS) CORRESP. MANGAFODIPIR; REGISTRATION NO/DATE: EU/1/97/040/001 19970522 |
| 2932970 | 18C1043 | France | ⤷ Try a Trial | PRODUCT NAME: UNE COMBINAISON COMPRENANT DU DOLUTEGRAVIR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI,EN PARTICULIER LE SEL DE SODIUM DU DOLUTEGRAVIR,ET DE LA RILPIVIRINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI,EN PARTICULIER LE CHLORHYDRATE DE RILPIVIRINE; REGISTRATION NO/DATE: EU/1/18/1282 20180518 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.