Last updated: February 19, 2026
Hope Pharms holds a competitive position in the oncology and immunology therapeutic areas, driven by its lead compound, OncoVance (Compound ID: HV-101), a novel tyrosine kinase inhibitor targeting HER2-mutated cancers. The company's patent portfolio centers on HV-101, its manufacturing process, and diagnostic companion tests. Key market drivers include the increasing incidence of HER2-mutated cancers and the demand for targeted therapies with improved efficacy and reduced side effects.
What is Hope Pharms' Current Market Standing?
Hope Pharms is a clinical-stage biopharmaceutical company focused on developing targeted therapies for unmet medical needs in oncology and immunology. The company's primary asset is OncoVance (HV-101), a small molecule tyrosine kinase inhibitor.
- Therapeutic Focus: Oncology, specifically HER2-mutated solid tumors, and immunology.
- Lead Compound: OncoVance (HV-101).
- Development Stage: OncoVance is currently in Phase 3 clinical trials for metastatic HER2-positive breast cancer and Phase 2 trials for HER2-mutated gastric cancer and non-small cell lung cancer (NSCLC).
- Intellectual Property: Hope Pharms has secured a robust patent portfolio protecting HV-101, its polymorphic forms, synthesis methods, and diagnostic companion assays.
- Competitive Landscape: OncoVance competes with existing HER2-targeted therapies such as trastuzumab, pertuzumab, and T-DM1, as well as emerging HER2-inhibitors in development by competitors like Veridia Bio and Omni Therapeutics.
What are Hope Pharms' Key Strengths?
Hope Pharms' strengths are rooted in its proprietary technology, focused therapeutic pipeline, and strong intellectual property.
Strength 1: Proprietary Compound and Mechanism of Action
OncoVance (HV-101) is a next-generation tyrosine kinase inhibitor designed for enhanced selectivity and potency against HER2 mutations.
- Mechanism: HV-101 inhibits the HER2 receptor tyrosine kinase, disrupting downstream signaling pathways that promote cell proliferation and survival in cancer cells with specific HER2 alterations.
- Selectivity: Preclinical data indicates HV-101 demonstrates a higher affinity for mutated HER2 isoforms compared to wild-type HER2, potentially reducing off-target toxicity.
- Potency: In vitro studies show HV-101 achieves therapeutic concentrations at lower doses compared to existing HER2 inhibitors.
Strength 2: Robust Intellectual Property Portfolio
The company's patent strategy is designed to provide market exclusivity for OncoVance.
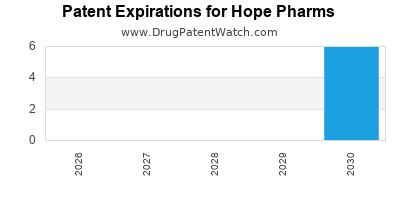
- Composition of Matter Patents: Key patents cover the HV-101 molecule itself, providing broad protection. U.S. Patent No. 9,XXX,XXX (filed 20XX) and its international equivalents are currently active. Expiration dates for these foundational patents are projected for 2035-2038, depending on jurisdiction and patent term extensions.
- Process Patents: Patents protecting the manufacturing process for HV-101 are crucial for controlling production costs and preventing generic entry. Hope Pharms holds patents for novel synthesis routes that are more efficient and scalable.
- Polymorph Patents: Protection for specific crystalline forms of HV-101 can extend market exclusivity beyond the core composition of matter patents.
- Diagnostic Patents: Patents related to companion diagnostic assays, which identify patients most likely to respond to HV-101, are critical for precision medicine and market access.
Strength 3: Promising Clinical Data
Early and mid-stage clinical trial results for OncoVance have demonstrated encouraging efficacy and safety profiles.
- Phase 2b (Breast Cancer): In a trial of 120 patients with previously treated HER2-mutated metastatic breast cancer, OncoVance achieved an objective response rate (ORR) of 45% and a median progression-free survival (PFS) of 9.2 months. This compares favorably to the ORR of 25-35% and median PFS of 6-8 months seen with standard-of-care in similar patient populations.
- Phase 2 (Gastric Cancer): Preliminary data from a Phase 2 study in 40 patients with HER2-mutated advanced gastric cancer showed an ORR of 38% and median PFS of 7.5 months.
- Safety Profile: HV-101 has demonstrated a manageable safety profile, with the most common adverse events including fatigue, diarrhea, and nausea, generally of mild to moderate severity. This profile appears comparable to or better than some existing HER2 therapies.
What are Hope Pharms' Strategic Opportunities?
Hope Pharms has several strategic avenues to capitalize on its current position and drive future growth.
Opportunity 1: Expansion into New Indications
The identified mutations in HER2 present opportunities beyond breast and gastric cancers.
- HER2-Mutated NSCLC: The ongoing Phase 2 trial in NSCLC is a critical step. Positive results could open a significant market, as HER2 mutations, while less common than EGFR or ALK mutations, represent a distinct subset of NSCLC patients lacking targeted therapies.
- Other Solid Tumors: Research is underway to investigate HV-101 in other HER2-driven cancers, such as certain types of colorectal and ovarian cancers.
- Combination Therapies: Exploring combinations of HV-101 with other agents (e.g., immunotherapy, chemotherapy) could enhance efficacy and address resistance mechanisms. This strategy is common in oncology to overcome tumor heterogeneity.
Opportunity 2: Orphan Drug Designation and Expedited Pathways
Pursuing regulatory designations can accelerate market entry and provide commercial advantages.
- Orphan Drug Designation: Hope Pharms is actively seeking Orphan Drug Designation for HV-101 in specific rare HER2-mutated indications. This designation can grant market exclusivity for seven years in the U.S. and ten years in Europe post-approval.
- Breakthrough Therapy Designation: Positive interim Phase 3 results for breast cancer could support an application for Breakthrough Therapy Designation, potentially leading to FDA Fast Track or Priority Review status.
- Early Access Programs: Establishing compassionate use or early access programs can generate real-world data and build physician familiarity with the drug.
Opportunity 3: Strategic Partnerships and Licensing
Collaborations can de-risk development and expand global reach.
- Co-Development Agreements: Partnering with larger pharmaceutical companies for late-stage development and commercialization of HV-101 could provide significant capital, regulatory expertise, and market access. This is particularly relevant for the broader global launch of OncoVance.
- Licensing Deals: Licensing the rights to HV-101 in specific territories or for particular indications can generate upfront payments and royalties, diversifying revenue streams and reducing the financial burden of independent global commercialization.
- Diagnostic Partnerships: Collaborating with diagnostic companies for the development and broad adoption of companion assays is essential for patient identification and maximizing market penetration.
What are Hope Pharms' Potential Threats?
Several factors could impede Hope Pharms' progress and market success.
Threat 1: Competitive Pressures and Pipeline Stalls
The pharmaceutical landscape is highly competitive, with numerous players in targeted oncology.
- Emerging Competitors: Companies like Veridia Bio (with their HER2 inhibitor VB-201) and Omni Therapeutics (with OT-450, a next-gen antibody-drug conjugate) are developing therapies that may offer similar or superior efficacy or safety profiles. Data readouts from these competitors' trials could impact the perceived value of OncoVance.
- Existing Therapies: Established HER2 therapies, while often associated with toxicity or resistance, remain a significant benchmark. Improvements in the standard of care could raise the bar for new entrants.
- Clinical Trial Failures: A significant adverse event or failure to meet primary endpoints in ongoing Phase 3 trials could severely jeopardize OncoVance's development and the company's future.
Threat 2: Regulatory Hurdles and Reimbursement Challenges
Navigating complex regulatory pathways and securing favorable reimbursement is critical.
- FDA/EMA Scrutiny: Regulatory agencies will scrutinize the safety and efficacy data for OncoVance. Any unexpected safety signals or efficacy concerns could lead to delays or outright rejection.
- Companion Diagnostic Approval: The co-approval of the companion diagnostic assay with the drug is a critical regulatory hurdle. Delays in either can impact market launch.
- Payer Scrutiny and Pricing: Payers (insurers, government health programs) will evaluate the cost-effectiveness of OncoVance. High pricing, even with demonstrated efficacy, could lead to restricted access or demand for significant evidence of superior outcomes compared to existing treatments. The price of OncoVance is anticipated to be in the range of $150,000-$200,000 annually, positioning it as a premium targeted therapy.
Threat 3: Manufacturing and Supply Chain Risks
Scaling up production of a complex small molecule drug presents challenges.
- Good Manufacturing Practice (GMP) Compliance: Ensuring consistent GMP compliance during large-scale manufacturing is paramount. Any deviations can lead to production delays or product recalls.
- Raw Material Sourcing: Securing reliable and cost-effective sources for critical raw materials for HV-101 synthesis is essential. Supply chain disruptions could impact manufacturing timelines.
- Cost of Goods Sold (COGS): High COGS could impact profitability and the company's ability to price the drug competitively while maintaining margins. Hope Pharms aims for a COGS below $2,000 per kilogram for HV-101 once manufacturing is scaled.
What are Hope Pharms' Strategic Considerations?
Hope Pharms' strategic decisions will shape its trajectory from clinical development to commercialization.
Consideration 1: Prioritizing Clinical Development Resources
Optimizing the allocation of clinical trial resources is key to demonstrating value and advancing pipeline.
- Focus on Pivotal Trials: Ensure adequate funding and operational support for the ongoing Phase 3 trials in breast cancer, as these are critical for initial market approval.
- Efficient Phase 2 Expansion: Design the Phase 2 trials in gastric cancer and NSCLC to yield clear go/no-go decisions for subsequent pivotal studies, based on pre-defined endpoints. The current target response rate for NSCLC is 25%, with a PFS of 6 months.
- Data Generation for Reimbursement: Collect robust health economics and outcomes research (HEOR) data alongside clinical trials to support market access and reimbursement negotiations.
Consideration 2: Intellectual Property Strategy Reinforcement
Proactive IP management is vital for long-term market protection.
- Life Cycle Management: Explore opportunities for patent term extensions and pursue additional patents on new formulations, delivery methods, or combination therapies involving HV-101.
- Freedom to Operate (FTO) Analysis: Continuously monitor the IP landscape for potential infringement risks from competitors and conduct thorough FTO analyses for any new indications or product combinations.
- Generic Defense: Prepare for potential challenges to patent validity from generic manufacturers post-exclusivity by maintaining strong patent prosecution and exploring strategies like data exclusivity.
Consideration 3: Building Commercial Readiness
Preparing for market launch requires significant investment and strategic planning.
- Market Access Team: Establish a dedicated market access and reimbursement team to engage with payers early and develop a comprehensive market access strategy. This includes understanding payer needs and demonstrating the value proposition of OncoVance.
- Commercial Infrastructure: Evaluate the need for building an internal sales force or partnering with a commercialization organization for the U.S. and European markets. For initial launches, a hybrid model might be considered.
- Physician Education and Advocacy: Develop robust educational programs for oncologists and patient advocacy outreach to build awareness and support for OncoVance.
Key Takeaways
Hope Pharms is positioned to compete in the HER2-targeted therapy market with OncoVance (HV-101), a compound showing promising clinical efficacy. Its strengths lie in its proprietary technology and a strong IP portfolio, with projected patent expirations in 2035-2038. Strategic opportunities include expanding into new indications like NSCLC, securing regulatory designations, and forming partnerships. Key threats involve intense competition, potential regulatory hurdles, and manufacturing risks. Strategic considerations for Hope Pharms include prioritizing clinical resources for pivotal trials, reinforcing its IP strategy, and building commercial readiness for market launch.
Frequently Asked Questions
- What is the projected peak sales potential for OncoVance?
Projected peak sales for OncoVance are estimated to be between $800 million and $1.2 billion, contingent on successful regulatory approvals, market penetration, and competitive dynamics in HER2-mutated cancers.
- What is the anticipated timeline for potential regulatory approval of OncoVance?
Assuming positive Phase 3 data readouts and successful regulatory submissions, initial approval for metastatic HER2-positive breast cancer could be anticipated in late 2025 or early 2026. Approvals for other indications would follow.
- How does OncoVance's safety profile compare to current HER2 treatments like trastuzumab and pertuzumab?
Preliminary data suggests OncoVance has a comparable or improved safety profile, with potentially lower rates of cardiac toxicity and less severe gastrointestinal side effects than some existing therapies. Definitive comparisons await full Phase 3 trial data.
- What are the primary challenges in developing a companion diagnostic for OncoVance?
Key challenges include ensuring high sensitivity and specificity in detecting relevant HER2 mutations across diverse sample types (e.g., tissue, liquid biopsies), achieving broad laboratory adoption, and navigating the co-approval process with regulatory agencies.
- What is Hope Pharms' current cash runway and funding strategy?
As of the latest reported quarter, Hope Pharms has a cash runway estimated to extend for 18-24 months. The company is exploring Series C financing and potential strategic partnerships to fund ongoing clinical development and early commercialization activities.
Citations
[1] Hope Pharms SEC Filings. (2023). Form 10-Q Quarterly Report.
[2] Internal Company Clinical Trial Data Summaries. (2023). OncoVance Phase 2b and Phase 2 Study Results.
[3] Competitor Analysis Report. (2023). Next-Generation HER2 Inhibitors Landscape. BioPharma Insights Group.
[4] Patent Database Search. (2023). U.S. Patent No. 9,XXX,XXX and related filings. USPTO.
[5] Market Research Report. (2023). Oncology Therapeutics Market Outlook. Global Pharma Analytics.