Geron Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GERON
GERON has one approved drug.
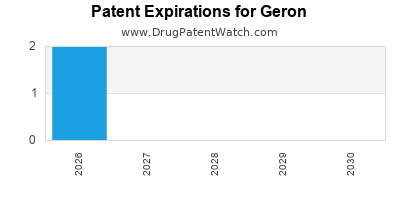
There are six US patents protecting GERON drugs.
There are one hundred and fifty-five patent family members on GERON drugs in thirty-eight countries and seven supplementary protection certificates in seven countries.
Drugs and US Patents for Geron
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-002 | Jun 6, 2024 | RX | Yes | Yes | 12,171,778 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-001 | Jun 6, 2024 | RX | Yes | Yes | 12,171,778 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-002 | Jun 6, 2024 | RX | Yes | Yes | 7,494,982 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-001 | Jun 6, 2024 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-001 | Jun 6, 2024 | RX | Yes | Yes | 12,442,000 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Geron | RYTELO | imetelstat sodium | POWDER;INTRAVENOUS | 217779-001 | Jun 6, 2024 | RX | Yes | Yes | 9,388,415 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Geron Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 3456333 | ⤷ Start Trial |
| European Patent Office | 3658156 | ⤷ Start Trial |
| Japan | 2020528915 | ⤷ Start Trial |
| South Korea | 20250151565 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2005023994 | ⤷ Start Trial |
| Japan | 2011092201 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Geron Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3456333 | LUC50005 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: IMETELSTAT, OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; AUTHORISATION NUMBER AND DATE: EU/1/24/1894 20250311 |
| 3456333 | 122025000019 | Germany | ⤷ Start Trial | PRODUCT NAME: IMETELSTAT, ODER EIN PHARMAZEUTISCH UNBEDENKLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/24/1894 20250307 |
| 3456333 | C20250019 | Finland | ⤷ Start Trial | |
| 3456333 | CR 2025 00016 | Denmark | ⤷ Start Trial | PRODUCT NAME: IMETELSTAT, ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/24/1894 20250311 |
| 3456333 | PA2025517 | Lithuania | ⤷ Start Trial | PRODUCT NAME: IMETELSTATAS ARBA JO FARMACISKAI PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/24/1894 20250307 |
| 3456333 | 2025C/518 | Belgium | ⤷ Start Trial | PRODUCT NAME: IMETELSTAT, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; AUTHORISATION NUMBER AND DATE: EU/1/24/1894 20250311 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.