Fresenius Kabi Usa Company Profile
✉ Email this page to a colleague
What is the competitive landscape for FRESENIUS KABI USA, and what generic alternatives to FRESENIUS KABI USA drugs are available?
FRESENIUS KABI USA has three hundred and one approved drugs.
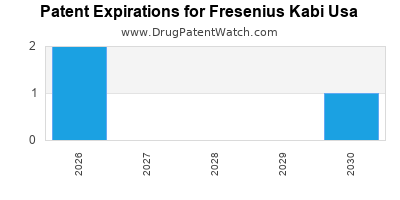
There are twenty-three US patents protecting FRESENIUS KABI USA drugs. There are two tentative approvals on FRESENIUS KABI USA drugs.
There are one hundred and twenty-nine patent family members on FRESENIUS KABI USA drugs in twenty-five countries and four hundred and sixty-two supplementary protection certificates in seventeen countries.
Summary for Fresenius Kabi Usa
| International Patents: | 129 |
| US Patents: | 23 |
| Tradenames: | 247 |
| Ingredients: | 184 |
| NDAs: | 301 |
| Patent Litigation for Fresenius Kabi Usa: | See patent lawsuits for Fresenius Kabi Usa |
| PTAB Cases with Fresenius Kabi Usa as petitioner: | See PTAB cases with Fresenius Kabi Usa as petitioner |
| PTAB Cases with Fresenius Kabi Usa as patent owner: | See PTAB cases with Fresenius Kabi Usa as patent owner |
Drugs and US Patents for Fresenius Kabi Usa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresenius Kabi Usa | SODIUM CHLORIDE 0.9% IN PLASTIC CONTAINER | sodium chloride | INJECTABLE;INJECTION | 207310-001 | Sep 19, 2017 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Kabi Usa | MAGNESIUM SULFATE IN PLASTIC CONTAINER | magnesium sulfate | INJECTABLE;INJECTION | 206485-001 | Mar 15, 2016 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Kabi Usa | NESACAINE-MPF | chloroprocaine hydrochloride | INJECTABLE;INJECTION | 009435-004 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Fresenius Kabi Usa | CALCITRIOL | calcitriol | INJECTABLE;INJECTION | 075836-002 | Dec 31, 2002 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Fresenius Kabi Usa | TERBUTALINE SULFATE | terbutaline sulfate | INJECTABLE;INJECTION | 076887-001 | May 26, 2004 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Kabi Usa | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 017029-004 | Approved Prior to Jan 1, 1982 | AP | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Kabi Usa | LEVOFLOXACIN IN DEXTROSE 5% IN PLASTIC CONTAINER | levofloxacin | INJECTABLE;INJECTION | 200674-003 | Jun 19, 2013 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Fresenius Kabi Usa
Paragraph IV (Patent) Challenges for FRESENIUS KABI USA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 2 mg/mL, 200 mL | ➤ Subscribe | 2015-09-03 |
| ➤ Subscribe | Injection | 1 mg/mL, 50 mL vials | ➤ Subscribe | 2011-12-16 |
| ➤ Subscribe | Injection | 2 mg/mL | ➤ Subscribe | 2011-06-22 |
| ➤ Subscribe | Injection | 10 mg/mL | ➤ Subscribe | 2011-11-04 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | Injection | 2 mg/mL, 100 mL | ➤ Subscribe | 2015-01-30 |
| ➤ Subscribe | Injection | 100 mg/mL, 2.5 mL vials | ➤ Subscribe | 2007-09-24 |
| ➤ Subscribe | Oral Solution | 5 mg/5mL | ➤ Subscribe | 2011-02-25 |
| ➤ Subscribe | Tablets | 2 mg, 4 mg, and 8 mg | ➤ Subscribe | 2013-08-05 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Injection | 2 mg/mL, 5 mg/mL and 10 mg/mL, 20 mL, 30 mL and 20 mL vials | ➤ Subscribe | 2006-11-13 |
International Patents for Fresenius Kabi Usa Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Portugal | 1507502 | ⤷ Try a Trial |
| Japan | 2016512455 | ⤷ Try a Trial |
| China | 100398084 | ⤷ Try a Trial |
| Germany | 10313760 | ⤷ Try a Trial |
| Austria | 469630 | ⤷ Try a Trial |
| Portugal | 1605891 | ⤷ Try a Trial |
| China | 102421426 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Fresenius Kabi Usa Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2822954 | 1890030-8 | Sweden | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BICTEGRAVIR SODIUM; REG. NO/DATE: EU/1/18/1289 20180625 |
| 1853250 | 37/2014 | Austria | ⤷ Try a Trial | PRODUCT NAME: PACLITAXEL, DAS ALS ALBUMIN GEBUNDENE NANOPARTIKEL FORMUIERT IST.; REGISTRATION NO/DATE: EU/1/07/428 (MITTEILUNG) (GEAENDERT DURCH C(2013) 9835) 20131230 |
| 0383579 | 97C0029 | Belgium | ⤷ Try a Trial | PRODUCT NAME: REMIFENTANILUM HYDROCHLORIDUM (= REMIFENTANILUM BASIS); NAT. REGISTRATION NO/DATE: 725 IS 146 F 12 19970408; FIRST REGISTRATION: DE 36.335.00.00 19960517 |
| 0350733 | SPC/GB03/034 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MOXIFLOXACIN AND PHARMACEUTICALLY USABLE HYDRATES AND ACID ADDITION SALTS THEREOF AND THE ALKALI METAL, ALKALINE EARTH METAL, SILVER AND GUANIDINIUM SALTS OF THE UNDERLYING CARBOXYLIC ACIDS AND THE RACEMATES THEREOF; REGISTERED: DE 45263.00.00 19990621; UK PL 000 10/0291 20030313 |
| 2822954 | 18C1035 | France | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE,EN PARTICULIER BICTEGRAVIR DE SODIUM; REGISTRATION NO/DATE: EU/1/18/1289 20180625 |
| 0480717 | 98C0025 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LOSARTAN POTASSIUM; HYDROCHLOROTHIAZIDE; NAT. REGISTRATION NO/DATE: NL 20 037 19950215; FIRST REGISTRATION: FR - NL 20 037 19950215 |
| 1853250 | 300673 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: PACLITAXEL, GEFORMULEERD ALS ALBUMINE-GEBONDEN NANODEELTJES; REGISTRATION NO/DATE: EU/1/07/428/001-002 20131220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.