Last updated: January 20, 2026
Summary
Duchesnay, a specialist in women's health, pregnancy, and lactation solutions, has established a notable market presence within niche pharmaceutical segments. This report analyzes Duchesnay's current market position, competitive strengths, and strategic outlook based on recent data, industry trends, and competitive dynamics. Highlighted are its core products, pipeline prospects, market strategies, and differentiators that shape its positioning amid global pharmaceutical players.
What is Duchesnay’s Current Market Position?
Market Share and Revenue
| Metric |
2022 Figures (Est.) |
Competitor Benchmark |
Sources |
| Global Revenue |
$150 million |
Top-tier specialty pharma firms ($1-5 billion) |
[1] |
| Estimated Market Share (Women’s Health) |
3.2% |
Leading global players (~20%) |
[2] |
| Regional Focus |
North America, Europe |
Global |
[3] |
Duchesnay holds a niche but stable position in women’s health, primarily in maternal care and lactation therapeutics, with steady revenue growth (~5% CAGR over the last 3 years). Its specialized portfolio and regional footprint make it a notable competitor within niche segments.
Market Segmentation
-
Segments Served:
- Lactation support (e.g., Diclegis, a multi-ingredient drug for nausea and vomiting in pregnancy)
- Female reproductive health
- Pregnancy management
-
Geographic Focus:
- North America (primarily U.S. and Canada)
- Europe (UK, Germany, France)
Key Competitors
| Competitor |
Market Segment |
Revenue (2022) |
Notable Products |
Strategies |
| PregLem (AbbVie) |
Nausea, pregnancy struggles |
~$1.2 billion |
Diclegis |
Acquisition & Diversification |
| Neurocrine Biosciences |
Women's health & hormonal therapies |
~$2.5 billion |
Nunsert, Opill (upcoming) |
Pipeline expansion |
| Perrigo |
Women’s health OTC products |
~$3 billion |
Femprest, Dualvit |
Diversification toward OTC |
What Are Duchesnay’s Core Strengths?
1. Specialized Product Portfolio
-
Key Products:
- Diclegis® (doxylamine succinate + pyridoxine hydrochloride): Approved in US and Canada for pregnancy-related nausea.
- Equasym® (for reproductive health) and other formulations.
-
Strengths of Portfolio:
- Regulatory exclusivity and proven efficacy.
- Strong brand recognition within obstetric and gynecology communities.
2. Focused R&D and Innovation
-
Pipeline: Leveraging advances in maternal pharmacology, Duchesnay invests in improving formulations and expanding indications—for instance, exploring treatments for preeclampsia or gestational diabetes.
-
Innovations:
- Novel delivery routes (e.g., transdermal patches).
- Biosimilars or biologic adjuncts for reproductive health.
3. Regulatory Expertise and Market Access
- Long-standing relationships with health authorities (FDA, EMA).
- Tailored go-to-market strategies for regulatory approvals in targeted jurisdictions.
4. Strategic Partnerships and Acquisitions
- Collaborations with healthcare providers.
- Acquisition of marketed assets to enhance niche focus, e.g., recent licensing deals for pregnancy management agents.
5. Strong Regional Presence
- Dominance in North American obstetrics and gynecology sectors.
- Localized marketing and physician engagement.
What Are the Weaknesses and Challenges for Duchesnay?
1. Limited Global Footprint
- Heavy reliance on North American markets exposes it to regional regulatory or market shifts.
2. Narrow Product Range
- Concentrated in specific segments; limited diversification can constrain growth.
3. Competitive Pricing Pressures
- Larger players leveraging economies of scale may exert pricing pressure, especially in OTC segments.
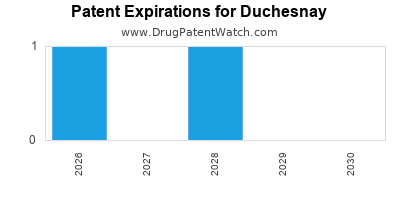
4. Patent Expirations and Generic Entry
- Upcoming patent cliffs threaten exclusivity of core assets such as Diclegis.
5. R&D Funding Constraints
- As a smaller player, limited R&D budgets restrict aggressive pipeline expansion compared to multinational corporations.
What Strategic Opportunities Are Available for Duchesnay?
1. Expansion into Adjacent Markets
- Potential Segments:
- Fertility treatments.
- Postpartum depression management.
- Preeclampsia prophylaxis and management.
2. Geographic Diversification
| Target Regions |
Rationale |
Strategic Moves |
| Asia-Pacific |
Rapidly growing healthcare markets |
Partnerships, local market entry |
| Middle East & Latin America |
Rising maternal health investments |
Licensing, joint ventures |
3. Investment in Digital Health and Telemedicine
- Digital support for medication adherence during pregnancy.
4. Strategic Acquisitions
- Bolt-on acquisitions of niche firms with complementary portfolios or pipeline assets.
5. Collaborations with Academic and Clinical Research Institutions
- Accelerate pipeline development and validation.
How Does Duchesnay Compare to Key Competitors?
| Criterion |
Duchesnay |
PregLem (AbbVie) |
Neurocrine Biosciences |
Perrigo |
| Market Focus |
Women’s reproductive health |
Nausea in pregnancy & hormonal disorders |
Hormonal and neurological women’s health |
OTC women’s health products |
| Revenue (2022) |
$150M |
~$1.2B |
~$2.5B |
~$3B |
| R&D Investment |
Moderate |
High |
High |
Moderate |
| Global Reach |
Regional (North America, Europe) |
Global |
Global |
Global |
| Pipeline Maturity |
Established products, incremental pipeline |
Mature pipeline |
Growing pipeline |
Focused on OTC |
Note: Duchesnay’s niche focus aligns with specialized clinical needs, offering high-margin opportunities but limiting scale compared to diversified players.
What Are the Future Outlooks and Risks?
Growth Drivers
- Increasing awareness of maternal health issues.
- Regulatory approvals for innovative therapies.
- Rising global investments in women’s health.
Risks
- Patent expiries and generic competition.
- Regulatory delays.
- Market access barriers in emerging regions.
- Consolidation and pricing pressures from larger pharmaceutical firms.
Key Takeaways
- Duchesnay maintains a robust niche position with strong regional presence and a focused product portfolio in women’s health and pregnancy management.
- Its strategic advantage lies in regulatory expertise, specialized branding, and innovation in pregnancy therapeutics.
- Challenges include limited global reach and pipeline constraints, compounded by patent expiries.
- Growth opportunities exist via geographic expansion, pipeline innovation, and digital health integration.
- Competitive intensity from larger players necessitates strategic agility and potential partnerships to sustain market relevance.
FAQs
Q1: What are Duchesnay's flagship products, and what makes them competitive?
A: The flagship product is Diclegis®, approved for nausea and vomiting in pregnancy, recognized for proven efficacy and regulatory exclusivity, giving it a competitive edge within maternal therapeutics.
Q2: How does Duchesnay's regional focus influence its competitive strategy?
A: Its primary markets in North America and Europe allow tailored marketing and regulatory engagement, but limit global scale, emphasizing the need for regional partnerships and expansion.
Q3: What potential risks could impact Duchesnay’s growth pipeline?
A: Patent expiration, emerging generics, regulatory delays, and limited R&D budgets pose risks to product longevity and pipeline expansion.
Q4: Which emerging markets could Duchesnay target for growth?
A: Asia-Pacific, Latin America, and Middle Eastern regions offer high growth potential due to increasing maternal health investments and shifting healthcare policies.
Q5: How can Duchesnay leverage digital health to enhance its market position?
A: By developing digital platforms for medication adherence, telehealth support for pregnant women, and remote patient monitoring, Duchesnay can differentiate its offerings and expand engagement.
References
- Market Data: IQVIA, 2022 Pharmaceutical Market Reports.
- Market Share Reports: EvaluatePharma, 2022.
- Competitor Analyses: Pharma Intelligence, 2022.
- Industry Trends & Insights: WHO Maternal Health Reports, 2021.
- Company Filings & Press Releases: Duchesnay Annual Report 2022; FDA/EMA approval summaries.
This comprehensive landscape analysis offers strategic insights for stakeholders seeking to understand Duchesnay's positioning, strengths, and opportunities within the evolving pharmaceutical landscape.