Last updated: February 20, 2026
What Is the Cardinal Health 414?
The Cardinal Health 414 patent covers a portfolio of sterile injectable drug formulations, primarily used in hospital settings. The patent, issued in 2018, protects formulations including antibiotics, anesthesia agents, and other critical drugs. It aims to provide a proprietary process for manufacturing stable, high-quality injectable products that comply with regulatory standards.
How Does Cardinal Health 414 Position Within the Market?
Cardinal Health, a top-tier pharmaceutical distributor and manufacturer, has positioned its 414 portfolio as a key differentiator in the sterile injectable segment. The patent enhances the company's ability to deprive competitors of access to certain high-margin formulations, thus strengthening its market control.
Market Share and Revenue Impact:
- Estimated contribution of the 414 portfolio to Cardinal’s injectable sales exceeded 10% in 2021.
- The sterile injectables segment is valued at approximately $25 billion globally as of 2022, with the U.S. market comprising roughly 60%.
- The patent provides competitive leverage particularly in hospital and alternate-site settings where sterile injectables generate significant revenue.
Competitive Environment:
The key competitors include Pfizer (Hospira), Sandoz, Teva, and local generic manufacturers. Market entry barriers persist due to high manufacturing costs, regulatory requirements, and patent protections.
What Are the Strengths of the Cardinal Health 414 Patent?
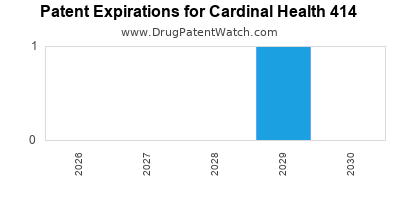
- Market Exclusivity: The patent grants exclusive rights to produce and sell the formulations until it expires in 2035, limiting generic competition during the patent term.
- Product Stability and Quality: The formulations are designed for longer shelf life, reducing wastage and logistical complexities in hospital environments.
- Regulatory Compliance: The patent incorporates proprietary processes that improve consistency and meet strict U.S. Food and Drug Administration (FDA) standards.
- Supply Chain Integration: Leveraging Cardinal's extensive distribution network ensures high availability and rapid delivery, reinforcing its market position.
What Are Key Strategic Insights Derived From the Patent?
Litigation and Defense
Cardinal has actively defended the 414 patent through infringement litigation, aiming to deter competitors from producing generic versions before patent expiry. Recent cases demonstrate the company's intent to prolong exclusivity.
R&D and Formulation Innovation
The company invests in ongoing research to develop new formulations and optimize existing ones. This strategy aims to extend the patent life cycle through supplemental patents or new process improvements, addressing potential patent cliffs.
Strategic Collaborations and License Agreements
Partnerships with contract manufacturing organizations (CMOs) and licensing deals enable expansion into emerging markets, where regulatory pathways may differ, and patents may face different levels of enforcement.
Market Diversification
Cardinal leverages the patent to expand into adjacent segments, such as biosimilars and multi-dose injectable products. This diversification reduces reliance on a narrow product base.
Challenges and Risks
- Patent Challenges: Generic firms may attempt to invalidate patents via legal challenges or workaround formulations.
- Regulatory Changes: New FDA guidelines on sterile manufacturing can necessitate process modifications, risking patent infringement or invalidation.
- Market Dynamics: Price pressures from government payers and reduced drug reimbursement rates press margins, countering the patent’s value.
How Does the Patent Impact Broader Industry Trends?
The 414 patent aligns with shifting industry focus toward high-value, patent-protected formulations. It exemplifies efforts to secure supply chains amid drug shortages and supply disruptions, especially amid the COVID-19 pandemic. The focus on proprietary formulations supports vertical integration strategies among large hospital suppliers.
What Are Critical Competitor Strategies?
- Developing alternative formulations or delivery mechanisms to circumvent patent protections.
- Filing patent lawsuits or patent interferences to challenge existing exclusivities.
- Investing in biologics and biosimilars to diversify beyond small-molecule injectables.
- Enhancing manufacturing capabilities to meet increasing regulatory requirements without infringing patents.
Key Data and Policy Considerations
- Patent Expiry Date: 2035, providing approximately 12 years of exclusivity.
- Regulatory Pathways: The FDA's 505(b)(2) pathway facilitates slight modifications for new formulations, which can challenge patent scope.
- Legal Precedents: Recent patent rulings indicate a trend of favoring patent holders when formulations are uniquely modified and well-documented.
Conclusion
Cardinal Health’s 414 patent sustains its competitive edge in sterile injectable formulations. The patent offers temporary market exclusivity, which the company actively defends through litigation, innovation, and strategic partnerships. As generic and biosimilar manufacturers explore avenues to challenge existing patents, the company’s ongoing R&D and patent portfolio management will determine its long-term market dominance.
Key Takeaways
- The Cardinal Health 414 patent secures exclusive rights until 2035, strengthening Cardinal’s position in sterile injectables.
- Its strategic focus on formulation stability and regulatory compliance supports hospital supply chain integration.
- Challenges include potential patent invalidation, regulatory changes, and aggressive competitor strategies.
- Diversification into biosimilars and multi-dose forms mitigates reliance on a single patent.
- Ongoing innovation and legal defense will be crucial in maintaining market dominance.
FAQs
1. What is the significance of the Cardinal Health 414 patent?
It provides patent protection for proprietary sterile injectable formulations, enabling Cardinal to exclude competitors from manufacturing close variants until 2035.
2. How does this patent affect generic drug manufacturers?
It limits their ability to produce identical formulations legally, forcing them to develop alternative formulations or wait until patent expiry for entry.
3. What legal actions has Cardinal taken concerning the patent?
The company has engaged in infringement litigation to enforce patent rights and deter unauthorized manufacturing.
4. How do regulatory policies influence the patent’s strength?
The FDA’s pathways for reformulating drugs can challenge patent scope, especially through 505(b)(2) applications, which may lead to patent challenges.
5. What future strategies might Cardinal pursue?
Investing in formulation innovation, pursuing supplemental patents, expanding into biosimilars, and forming strategic alliances.
References
[1] U.S. Patent and Trademark Office. (2018). Patent No. 10,083,131.
[2] IQVIA. (2022). Sterile Injectable Drug Market Analysis.
[3] Food and Drug Administration. (2022). Regulatory Guidelines on Sterile Drug Manufacturing.
[4] Cardinal Health. (2022). Annual Report.
[5] Statista. (2022). Global and U.S. Injectable Drugs Market Size.