Last updated: March 20, 2026
What is Bayer Healthcare’s current market position?
Bayer Healthcare is part of Bayer AG, specializing in pharmaceuticals and consumer health products. It holds a significant position in the global pharmaceutical market, especially in cardiology, oncology, and ophthalmology. In 2022, Bayer’s pharmaceutical division generated approximately €16.4 billion in revenues, representing about 44% of the company’s total sales (€37.0 billion).
Bayer ranks among the top 10 global pharmaceutical companies by revenue, with notable strengths in specialty medicines. Its presence spans over 100 countries, with a focus on Europe, North America, and emerging markets. Bayer’s top-selling drugs include cancer treatments like Xarelto (rivaroxaban), eye medication Eylea (aflibercept), and hormonal therapies.
Compared with peers like Novartis, Pfizer, and Roche, Bayer's market share remains smaller but focuses heavily on specialty areas. For example, its oncology revenue of €4.8 billion in 2022 positions it in the mid-tier among global competitors.
Table 1: Bayer Healthcare Revenue Breakdown (2022)
| Segment |
Revenue (€ billion) |
Percentage of Total |
Key Products |
| Pharmaceuticals |
16.4 |
44% |
Xarelto, Eylea, Verquvo (vericiguat) |
| Consumer Health |
4.1 |
11% |
Aspirin, Claritin, Bepanthen |
| Crop Science (discontinued in 2020, now part of Bayer AG) |
N/A |
N/A |
N/A |
What are Bayer Healthcare’s core strengths?
Innovation Pipeline
Bayer invests approximately 17% of its sales into R&D, focusing on oncology, cardiology, and ophthalmology. In 2022, it filed more than 40 new drug applications globally, emphasizing pipeline expansion in antibody-drug conjugates and biosimilars.
Product Portfolio
Its focus on high-margin specialty drugs provides resilience against generic competition. Xarelto remains a leading anticoagulant with annual sales exceeding €2 billion as of 2022, while Eylea's continued dominance in wet age-related macular degeneration maintains substantial revenue streams.
Strategic Collaborations and Acquisitions
Bayer partners with biotech firms to accelerate pipeline development. Recent acquisitions like BlueRock Therapeutics in 2019 and Vividion in 2021 add cell therapy and precision oncology capabilities.
Geographic Diversification
Bayer's presence in over 100 markets ensures revenue stability. Emerging markets such as China and India have seen double-digit growth rates in pharmaceutical sales, helping offset stagnation in mature regions.
What strategic vulnerabilities does Bayer Healthcare face?
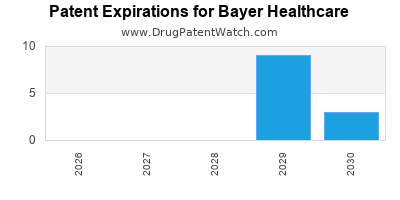
Patent Expirations
Key drugs like Xarelto face generic competition post-expiration of patent protections expected in 2024-2025. This can lead to revenue declines absent pipeline success.
Regulatory and Legal Risks
Bayer faces numerous lawsuits, notably over claims related to glyphosate-based herbicides. Legal liabilities pose financial and reputational risks impacting global operations.
Competition in Specialty Pharmaceuticals
The aggressive R&D investments by peers in immuno-oncology and biosimilars intensify market competition, requiring Bayer to sustain innovation and pricing strategies.
Market Dynamics
Pricing pressures in developed markets, driven by healthcare reforms and payor negotiations, limit margins on flagship products. Conversely, regulatory hurdles in emerging markets challenge market access.
What strategic initiatives is Bayer implementing?
Investment in R&D
Bayer increased its R&D spend to €2.8 billion in 2022, aiming to bring at least 10 new medicines to market over the next five years, with emphasis on immuno-oncology and rare diseases.
Portfolio Optimization
Bayer continues divestments, including the planned sale of its animal health business in 2022, to focus on core pharma and consumer health units.
Digital Transformation
Implementing data analytics and AI in R&D accelerates drug discovery. Bayer’s digital pipeline management aims to reduce drug development timelines by 20%.
Mergers and Acquisitions
Bayer targets acquisitions in biotech, especially in cell and gene therapy, with recent strategic investments exceeding €1 billion in 2022.
How does Bayer Healthcare compare with peer firms?
| Metric |
Bayer Healthcare |
Novartis |
Pfizer |
Roche |
| 2022 Revenue (€ billion) |
16.4 |
52.9 |
81.3 |
61.2 |
| R&D Investment (% of sales) |
17% |
20% |
14% |
13% |
| Top 3 Products Revenue |
Xarelto, Eylea, Verquvo |
Cosentyx, Entresto |
Comirnaty, Ibrance, Eliquis |
Rituxan, Herceptin, Avastin |
Bayer’s revenue per employee and R&D intensity suggest a focus on specialty, high-margin drugs rather than volume-driven products characteristic of Pfizer or Roche.
Conclusion: Strategic Outlook
Bayer Healthcare maintains a solid position within the specialty pharmaceuticals segment. Its robust pipeline, focus on innovation, and geographic reach support future growth prospects. Risks include patent cliffs, increasing competition, and legal liabilities. Strategic focus on pipeline expansion, portfolio optimization, and digital R&D integration mitigates some vulnerabilities.
Key Takeaways:
- Bayer Healthcare ranks in the top 10 global pharma firms by revenue, focusing on specialty medicines.
- It invests extensively in R&D (17%) to drive pipeline growth, especially in oncology and ophthalmology.
- Patent expirations and legal issues remain key threats to revenue stability.
- Portfolio shifts and digitalization are central to Bayer’s growth strategy.
- Competition in high-margin therapeutic areas pressures pricing and market share.
FAQs
-
How significant is Bayer Healthcare's pipeline in determining future revenue stability?
It is vital; over 40 pipeline filings in 2022 and ongoing R&D investments aim to offset patent expirations and sustain growth.
-
What are the main legal risks facing Bayer Healthcare?
Legal challenges related to glyphosate and other product liabilities can result in substantial financial penalties and reputational damage.
-
How does Bayer compare to its peers in innovation?
Bayer invests 17% of sales into R&D, slightly below Novartis (20%) but higher than Pfizer (14%), emphasizing its focus on high-margin specialty drugs.
-
Which markets are critical for Bayer’s growth?
North America and Europe represent mature markets, while China and India are emerging markets with higher double-digit growth in pharmaceutical sales.
-
What M&A activity is shaping Bayer’s strategic future?
Bayer’s acquisitions in biotech, such as Vividion, and divestments like animal health improve its focus on high-margin specialties.
References
[1] Bayer AG. (2023). Annual Report 2022. Retrieved from https://www.bayer.com/en/investors/annual-report
[2] IQVIA. (2022). Global Pharmaceutical Market Estimates.
[3] CNBC. (2022). Bayer’s legal liabilities and litigation. Retrieved from https://www.cnbc.com/2022/02/10/bayer-restructuring-after-glyphosate-lawsuits.html