BRISTOL-MYERS SQUIBB Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BRISTOL-MYERS SQUIBB, and what generic alternatives to BRISTOL-MYERS SQUIBB drugs are available?
BRISTOL-MYERS SQUIBB has one approved drug.
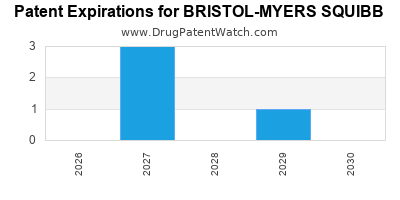
There are five US patents protecting BRISTOL-MYERS SQUIBB drugs.
There are eighty-one patent family members on BRISTOL-MYERS SQUIBB drugs in thirty-one countries and nineteen supplementary protection certificates in sixteen countries.
Summary for BRISTOL-MYERS SQUIBB
| International Patents: | 81 |
| US Patents: | 5 |
| Tradenames: | 1 |
| Ingredients: | 1 |
| NDAs: | 1 |
| Patent Litigation for BRISTOL-MYERS SQUIBB: | See patent lawsuits for BRISTOL-MYERS SQUIBB |
| PTAB Cases with BRISTOL-MYERS SQUIBB as patent owner: | See PTAB cases with BRISTOL-MYERS SQUIBB as patent owner |
Drugs and US Patents for BRISTOL-MYERS SQUIBB
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol-myers Squibb | DAKLINZA | daclatasvir dihydrochloride | TABLET;ORAL | 206843-001 | Jul 24, 2015 | DISCN | Yes | No | 9,421,192 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Bristol-myers Squibb | DAKLINZA | daclatasvir dihydrochloride | TABLET;ORAL | 206843-002 | Jul 24, 2015 | DISCN | Yes | No | 8,629,171 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Bristol-myers Squibb | DAKLINZA | daclatasvir dihydrochloride | TABLET;ORAL | 206843-002 | Jul 24, 2015 | DISCN | Yes | No | 9,421,192 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for BRISTOL-MYERS SQUIBB Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Hungary | E037802 | ⤷ Try a Trial |
| Denmark | 2784075 | ⤷ Try a Trial |
| Cyprus | 1119988 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for BRISTOL-MYERS SQUIBB Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2049522 | C 2015 003 | Romania | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR SI SARURILE ACCEPTABILE FARMACEUTIC ALEACESTUIA, IN SPECIAL DACLATASVIR DICLORHIDRAT; NATIONAL AUTHORISATION NUMBER: EU/1/14/939/001, EU/1/14/939/002, EU/1/14/939/003, EU/1/14/939/004; DATE OF NATIONAL AUTHORISATION: 20140822; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/14/939/001, EU/1/14/939/002, EU/1/14/939/003, EU/1/14/939/004; DATE OF FIRST AUTHORISATION IN EEA: 20140822 |
| 2049522 | 122015000009 | Germany | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR UND PHARMAZEUTISCH AKZEPTABLE SALZE DAVON, INSBESONDERE DACLATASVIR DIHYDROCHLORID; REGISTRATION NO/DATE: EU/1/14/939/001-004 20140822 |
| 2049522 | 92635 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR ET LEURS SELS PHARMACEUTIQUEMENT ACCEPTABLES , EN PARTICULIER DICHLORHYDRATE DE DACLATASVIR. FIRST REGISTRATION: 20140826 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.