ADHERA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ADHERA, and what generic alternatives to ADHERA drugs are available?
ADHERA has one approved drug.
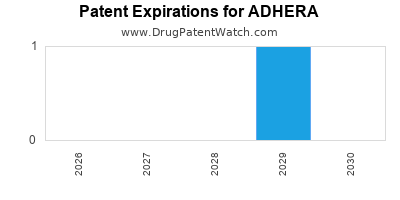
There is one US patent protecting ADHERA drugs.
There are twenty-seven patent family members on ADHERA drugs in twenty-seven countries and sixteen supplementary protection certificates in eight countries.
Drugs and US Patents for ADHERA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-002 | Jan 21, 2015 | RX | Yes | No | 7,846,961 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-003 | Jan 21, 2015 | RX | Yes | Yes | 7,846,961 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-001 | Jan 21, 2015 | RX | Yes | No | 7,846,961 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ADHERA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-003 | Jan 21, 2015 | 6,696,481 | ⤷ Try a Trial |
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-001 | Jan 21, 2015 | 6,696,481 | ⤷ Try a Trial |
| Adhera | PRESTALIA | amlodipine besylate; perindopril arginine | TABLET;ORAL | 205003-002 | Jan 21, 2015 | 6,696,481 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ADHERA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 3.5 mg/2.5 mg, 7 mg/5 mg and 14 mg/10 mg | ➤ Subscribe | 2016-11-04 |
International Patents for ADHERA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Hungary | E027898 | ⤷ Try a Trial |
| Eurasian Patent Organization | 200801777 | ⤷ Try a Trial |
| Poland | 1989182 | ⤷ Try a Trial |
| Montenegro | 02456 | ⤷ Try a Trial |
| Cyprus | 1117753 | ⤷ Try a Trial |
| Canada | 2644467 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for ADHERA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1507558 | C300528 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN ALISKIREN OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, AMLODIPINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN EN HYDROCHLOORTHIAZIDE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; NATL. REGISTRATION NO/DATE: EU/1/11/730/001-060 20111122; FIRST REGISTRATION: CH 61678 01-05 20110705 |
| 0502314 | SPC/GB11/010 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: THE COMBINATION OF A) TELMISARTAN, OPTIONALLY IN THE FORM OF PHARMACEUTICALLY ACCEPTABLE SALTS, AND B) AMLODIPINE, OPTIONALLY IN THE FORM OF PHARMACEUTICALLY ACCEPTABLE SALTS, ESPECIALLY AMLODIPINE BESYLATE; REGISTERED: UK EU/1/10/648/001 20101007; UK EU/1/10/648/002 20101007; UK EU/1/10/648/003 20101007; UK EU/1/10/648/004 20101007; UK EU/1/10/648/005 20101007; UK EU/1/10/648/006 20101007; UK EU/1/10/648/007 20101007; UK EU/1/10/648/008 20101007; UK EU/1/10/648/009 20101007; UK EU/1/10/648/010 20101007; UK EU/1/10/648/011 20101007; UK EU/1/10/648/012 20101007; UK EU/1/10/648/013 20101007; UK EU/1/10/648/014 20101007; UK EU/1/10/648/015 20101007; UK EU/1/10/648/016 20101007; UK |
| 0502314 | C300478 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN AMLODIPINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER AMLODIPINEBESILAAT; REGISTRATION NO/DATE: EU/1/10/648/001-028 20101007 |
| 1915993 | 92315 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON COMPRENANT ALISKIREN,OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLE,ET AMLODIPINE,OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLE |
| 0503785 | C300486 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN OLMESARTANMEDOXOMIL, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, AMLODIPINEBESYLAAT AND HYDROCHLOORTHIAZIDE; NATL REGISTRATION NO/DATE: RVG 106667, RVG 106671-74, RVG 106682-86 20101221; FIRST REGISTRATION: DE 79810.00.00-79814.00.00 20101216 |
| 1507558 | 2012/018 | Ireland | ⤷ Try a Trial | PRODUCT NAME: ALISKIREN OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, AMLODIPINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF AND HYDROCHLOROTHIAZIDE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; NAT REGISTRATION NO/DATE: EU/1/11/730/001-060 20111122; FIRST REGISTRATION NO/DATE: SWITZERLAND 6167801-6167805 20110705 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.