DEXAMETHASONE Drug Patent Profile
✉ Email this page to a colleague
When do Dexamethasone patents expire, and what generic alternatives are available?
Dexamethasone is a drug marketed by Alpharma Us Pharms, Anima, Chartwell Molecular, Lyne, Pharmobedient Cnsltg, Abraxis Pharm, Fresenius Kabi Usa, Hikma, Watson Labs, Alvogen, Amneal, Apotex, Bausch, Bionpharma, Chartwell Rx, Impax Labs, Larken Labs Inc, Novitium Pharma, Pangea, Phoenix Labs Ny, Prasco, Pvt Form, Roxane, Sun Pharm Industries, Upsher Smith, Whiteworth Town Plsn, Xspire Pharma, Zydus Pharms, Watson Labs Teva, Bel Mar, Dell Labs, Dr Reddys, Epic Pharma Llc, Eugia Pharma, Geneyork Pharms, Gland Pharma Ltd, Intl Medication, Luitpold, Lyphomed, Mylan Labs Ltd, Somerset, Somerset Theraps Llc, Teva Parenteral, West-ward Pharms Int, Wyeth Ayerst, Sola Barnes Hind, Bausch And Lomb, and Sandoz. and is included in ninety-six NDAs.
The generic ingredient in DEXAMETHASONE is dexamethasone acetate. There are thirty-nine drug master file entries for this compound. Additional details are available on the dexamethasone acetate profile page.
Summary for DEXAMETHASONE
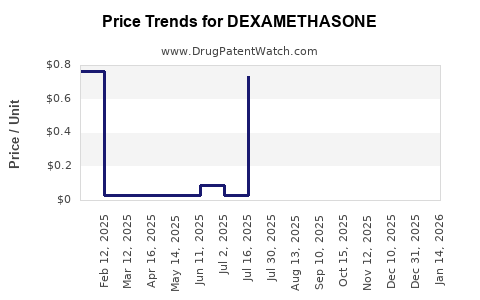
See drug prices for DEXAMETHASONE
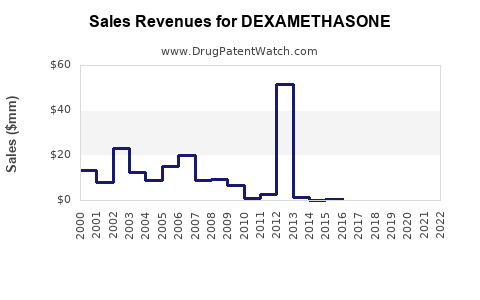
Recent Clinical Trials for DEXAMETHASONE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Dipenkumar Modi | Phase 2 |
| National Trauma Center | N/A |
| Genmab | Phase 2 |
Pharmacology for DEXAMETHASONE
| Drug Class | Corticosteroid |
| Mechanism of Action | Corticosteroid Hormone Receptor Agonists |
Medical Subject Heading (MeSH) Categories for DEXAMETHASONE
Anatomical Therapeutic Chemical (ATC) Classes for DEXAMETHASONE
US Patents and Regulatory Information for DEXAMETHASONE
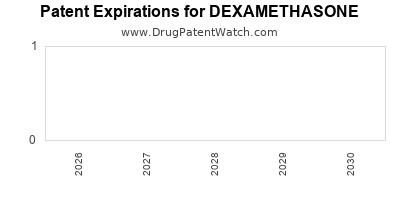
DEXAMETHASONE is protected by zero US patents and one FDA Regulatory Exclusivity.
FDA Regulatory Exclusivity protecting DEXAMETHASONE
COMPETITIVE GENERIC THERAPY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zydus Pharms | DEXAMETHASONE | dexamethasone | TABLET;ORAL | 216282-004 | Feb 7, 2024 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Teva Parenteral | DEXAMETHASONE SODIUM PHOSPHATE | dexamethasone sodium phosphate | INJECTABLE;INJECTION | 081125-001 | Aug 31, 1990 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Novitium Pharma | DEXAMETHASONE | dexamethasone | TABLET;ORAL | 217696-001 | May 9, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for DEXAMETHASONE
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AbbVie Deutschland GmbH & Co. KG | Ozurdex | dexamethasone | EMEA/H/C/001140 Ozurdex is indicated for the treatment of adult patients with macular oedema following either branch retinal-vein occlusion (BRVO) or central retinal-vein occlusion (CRVO).Ozurdex is indicated for the treatment of adult patients with inflammation of the posterior segment of the eye presenting as noninfectious uveitis.Ozurdex is indicated for the treatment of adult patients with visual impairment due to diabetic macular oedema (DME) who are pseudophakic or who are considered insufficiently responsive to, or unsuitable for non-corticosteroid therapy. |
Authorised | no | no | no | 2010-07-26 | |
| THERAVIA | Neofordex | dexamethasone | EMEA/H/C/004071 Treatment of multiple myeloma. |
Authorised | no | no | no | 2016-03-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |


