CYANOCOBALAMIN Drug Patent Profile
✉ Email this page to a colleague
When do Cyanocobalamin patents expire, and what generic alternatives are available?
Cyanocobalamin is a drug marketed by Abraxis Pharm, Am Regent, Dell Labs, Dr Reddys, Epic Pharma Llc, Eugia Pharma, Fresenius Kabi Usa, Gland Pharma Ltd, Luitpold, Lyphomed, Mylan Institutional, Mylan Labs Ltd, Sagent Pharms Inc, Sandoz, Sanofi Aventis Us, Sola Pharms, Solopak, Somerset Theraps Llc, Vitruvias Therap, Warner Chilcott, Watson Labs, West-ward Pharms Int, Wyeth Ayerst, Xiromed, Zydus Pharms, Lupin, Padagis Israel, and West Ward. and is included in twenty-nine NDAs.
The generic ingredient in CYANOCOBALAMIN is cyanocobalamin. There are nine drug master file entries for this compound. Twenty-three suppliers are listed for this compound. Additional details are available on the cyanocobalamin profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Cyanocobalamin
A generic version of CYANOCOBALAMIN was approved as cyanocobalamin by PADAGIS ISRAEL on September 9th, 2020.
Summary for CYANOCOBALAMIN
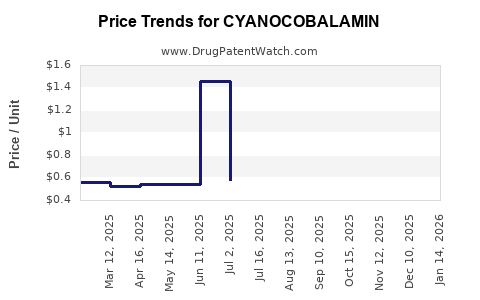
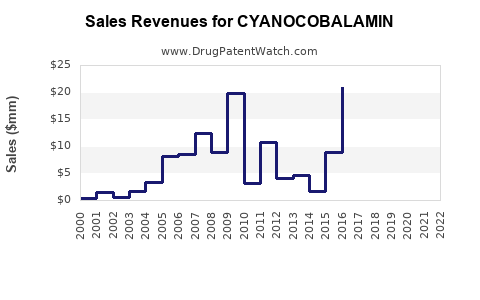
See drug prices for CYANOCOBALAMIN
Recent Clinical Trials for CYANOCOBALAMIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Fundação Educacional Serra dos Órgãos | Phase 4 |
| University of Kansas Medical Center | Phase 2 |
| Tanta University | Phase 3 |
Pharmacology for CYANOCOBALAMIN
| Drug Class | Vitamin B12 |
Anatomical Therapeutic Chemical (ATC) Classes for CYANOCOBALAMIN
Paragraph IV (Patent) Challenges for CYANOCOBALAMIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| NASCOBAL | Nasal Spray | cyanocobalamin | 500 mcg/spray | 021642 | 1 | 2017-04-28 |
US Patents and Regulatory Information for CYANOCOBALAMIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Xiromed | CYANOCOBALAMIN | cyanocobalamin | INJECTABLE;INJECTION | 215046-001 | Aug 20, 2021 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Watson Labs | CYANOCOBALAMIN | cyanocobalamin | INJECTABLE;INJECTION | 083120-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Zydus Pharms | CYANOCOBALAMIN | cyanocobalamin | INJECTABLE;INJECTION | 214655-001 | Apr 15, 2022 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| West-ward Pharms Int | CYANOCOBALAMIN | cyanocobalamin | INJECTABLE;INJECTION | 080515-002 | Approved Prior to Jan 1, 1982 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Abraxis Pharm | CYANOCOBALAMIN | cyanocobalamin | INJECTABLE;INJECTION | 080510-003 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


