CIALIS Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cialis, and when can generic versions of Cialis launch?
Cialis is a drug marketed by Lilly and is included in one NDA.
The generic ingredient in CIALIS is tadalafil. There are twenty-five drug master file entries for this compound. Forty-seven suppliers are listed for this compound. Additional details are available on the tadalafil profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Cialis
A generic version of CIALIS was approved as tadalafil by TEVA PHARMS USA on May 22nd, 2018.
Summary for CIALIS
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 108 |
| Clinical Trials: | 95 |
| Patent Applications: | 6,324 |
| Formulation / Manufacturing: | see details |
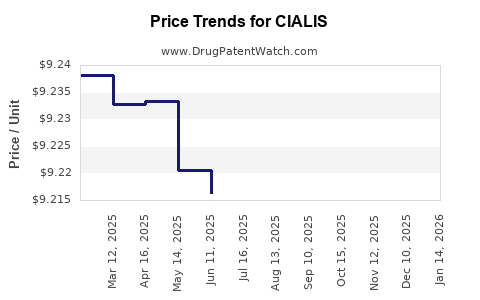
| Drug Prices: | Drug price information for CIALIS |
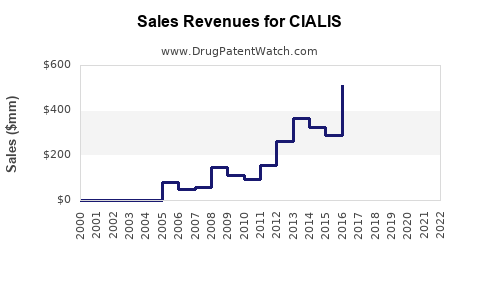
| Drug Sales Revenues: | Drug sales revenues for CIALIS |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CIALIS |
| What excipients (inactive ingredients) are in CIALIS? | CIALIS excipients list |
| DailyMed Link: | CIALIS at DailyMed |
Recent Clinical Trials for CIALIS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Arizona | Phase 2 |
| University of Florida | Phase 2/Phase 3 |
| Dongkook Pharmaceutical Co., Ltd. | Phase 3 |
Pharmacology for CIALIS
| Drug Class | Phosphodiesterase 5 Inhibitor |
| Mechanism of Action | Phosphodiesterase 5 Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for CIALIS
US Patents and Regulatory Information for CIALIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-004 | Jan 7, 2008 | AB1 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-003 | Nov 21, 2003 | AB1 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-001 | Nov 21, 2003 | AB1 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-002 | Nov 21, 2003 | AB1 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CIALIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-001 | Nov 21, 2003 | ⤷ Try a Trial | ⤷ Try a Trial |
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-001 | Nov 21, 2003 | ⤷ Try a Trial | ⤷ Try a Trial |
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-004 | Jan 7, 2008 | ⤷ Try a Trial | ⤷ Try a Trial |
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-002 | Nov 21, 2003 | ⤷ Try a Trial | ⤷ Try a Trial |
| Lilly | CIALIS | tadalafil | TABLET;ORAL | 021368-004 | Jan 7, 2008 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for CIALIS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Viatris Limited | Talmanco (previously Tadalafil Generics) | tadalafil | EMEA/H/C/004297 Talmanco is indicated in adults for the treatment of pulmonary arterial hypertension (PAH) classified as WHO functional class II and III, to improve exercise capacity. Efficacy has been shown in idiopathic PAH (IPAH) and in PAH related to collagen vascular disease. |
Authorised | yes | no | no | 2017-01-09 | |
| Eli Lilly Nederland B.V. | Tadalafil Lilly | tadalafil | EMEA/H/C/004666 Treatment of erectile dysfunction in adult males.In order for tadalafil to be effective, sexual stimulation is required.Tadalafil Lilly is not indicated for use by women.Treatment of the signs and symptoms of benign prostatic hyperplasia in adult males. |
Authorised | no | no | no | 2017-03-22 | |
| Eli Lilly Nederland B.V. | Adcirca (previously Tadalafil Lilly) | tadalafil | EMEA/H/C/001021 AdultsTreatment of pulmonary arterial hypertension (PAH) classified as WHO functional class II and III, to improve exercise capacity (see section 5.1).Efficacy has been shown in idiopathic PAH (IPAH) and in PAH related to collagen vascular disease.Paediatric populationTreatment of paediatric patients aged 2 years and above with pulmonary arterial hypertension (PAH) classified as WHO functional class II and III. |
Authorised | no | no | no | 2008-10-01 | |
| Eli Lilly Nederland B.V. | Cialis | tadalafil | EMEA/H/C/000436 Treatment of erectile dysfunction.In order for tadalafil to be effective, sexual stimulation is required.Cialis is not indicated for use by women. |
Authorised | no | no | no | 2002-11-12 | |
| Mylan Pharmaceuticals Limited | Tadalafil Mylan | tadalafil | EMEA/H/C/003787 Treatment of erectile dysfunction in adult males.In order for tadalafil to be effective, sexual stimulation is required.Tadalafil Mylan is not indicated for use by women. |
Authorised | yes | no | no | 2014-11-21 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for CIALIS
See the table below for patents covering CIALIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Peru | 00712001 | ⤷ Try a Trial | |
| Peru | 49495 | DERIVADO TETRACICLICO Y PROCESO PARA SU PREPARACION | ⤷ Try a Trial |
| European Patent Office | 1200092 | PRODUITS DE MEDICAMENTS A BASE DE BETA-CARBOLINE (BETA-CARBOLINE DRUG PRODUCTS) | ⤷ Try a Trial |
| Algeria | 3180 | Produits de médicaments à base de beta-carboline. | ⤷ Try a Trial |
| Poland | 315559 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CIALIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2101777 | 300813 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN TOEGEPAST IN COMBINATIEBEHANDELING MET TADALAFIL; REGISTRATION NO/DATE: EU/1/08/451 (C(2008) 1637) 20151125 |
| 2101777 | 2016C/032 | Belgium | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN EN COMBINAISON AVEC LE TADALAFIL; AUTHORISATION NUMBER AND DATE: EU/1/08/451 20151125 |
| 0740668 | C00740668/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: TADALAFILUM; REGISTRATION NUMBER/DATE: SWISSMEDIC 56018 04.05.2004 |
| 2101777 | 122016000039 | Germany | ⤷ Try a Trial | PRODUCT NAME: AMBRISENTAN IN VERWENDUNG IN DER KOMBINATIONSTHERAPIE MIT TADALAFIL; REGISTRATION NO/DATE: EU/1/08/451 20151120 |
| 0740668 | 03C0017 | France | ⤷ Try a Trial | PRODUCT NAME: TADALAFIL; REGISTRATION NO/DATE: EU/1/02/237/001-004 20021112 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


