TELMISARTAN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Telmisartan, and when can generic versions of Telmisartan launch?
Telmisartan is a drug marketed by Alembic, Amneal Pharms, Aurobindo Pharma, Cadila Pharms Ltd, Chartwell Rx, Glenmark Pharms Ltd, Hetero Labs Ltd V, Hisun Pharm Hangzhou, Inventia, Jubilant Generics, Micro Labs, Mylan, Prinston Inc, Sandoz, Torrent, Zydus Pharms, Lupin Ltd, and Macleods Pharms Ltd. and is included in twenty-nine NDAs.
The generic ingredient in TELMISARTAN is hydrochlorothiazide; telmisartan. There are thirty-two drug master file entries for this compound. Nine suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; telmisartan profile page.
Summary for TELMISARTAN
| US Patents: | 0 |
| Applicants: | 18 |
| NDAs: | 29 |
| Finished Product Suppliers / Packagers: | 25 |
| Raw Ingredient (Bulk) Api Vendors: | 151 |
| Clinical Trials: | 270 |
| Patent Applications: | 4,542 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for TELMISARTAN |
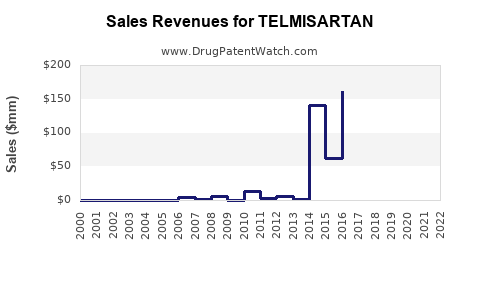
| Drug Sales Revenues: | Drug sales revenues for TELMISARTAN |
| What excipients (inactive ingredients) are in TELMISARTAN? | TELMISARTAN excipients list |
| DailyMed Link: | TELMISARTAN at DailyMed |
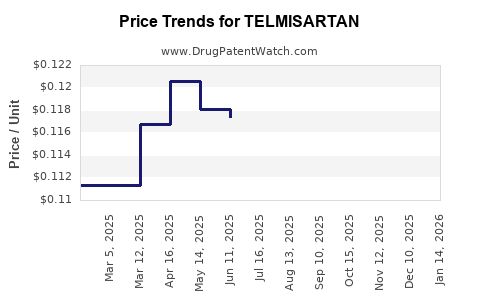
See drug prices for TELMISARTAN
Recent Clinical Trials for TELMISARTAN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| HK inno.N Corporation | Phase 1 |
| Michael Smith Foundation for Health Research | Early Phase 1 |
| Heart and Stroke Foundation of Canada | Early Phase 1 |
Pharmacology for TELMISARTAN
| Drug Class | Angiotensin 2 Receptor Blocker |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for TELMISARTAN
Paragraph IV (Patent) Challenges for TELMISARTAN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| MICARDIS | Tablets | telmisartan | 20 mg, 40 mg and 80 mg | 020850 | 1 | 2006-12-26 |
US Patents and Regulatory Information for TELMISARTAN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glenmark Pharms Ltd | TELMISARTAN AND HYDROCHLOROTHIAZIDE | hydrochlorothiazide; telmisartan | TABLET;ORAL | 202544-002 | Mar 4, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Sandoz | TELMISARTAN | telmisartan | TABLET;ORAL | 203867-002 | Nov 3, 2014 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Micro Labs | TELMISARTAN | telmisartan | TABLET;ORAL | 207016-002 | Oct 3, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Torrent | TELMISARTAN AND HYDROCHLOROTHIAZIDE | hydrochlorothiazide; telmisartan | TABLET;ORAL | 201192-001 | Feb 25, 2014 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for TELMISARTAN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Micardis | telmisartan | EMEA/H/C/000209 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type-2 diabetes mellitus with documented target-organ damage. |
Authorised | no | no | no | 1998-12-16 | |
| Krka, d.d., Novo mesto | Tolura | telmisartan | EMEA/H/C/001196 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease or peripheral arterial disease) or;type 2 diabetes mellitus with documented target organ damage. |
Authorised | yes | no | no | 2010-06-04 | |
| Teva B.V. | Telmisartan Teva Pharma | telmisartan | EMEA/H/C/002511 Treatment of essential hypertension in adults. |
Authorised | yes | no | no | 2011-10-03 | |
| Teva B.V. | Telmisartan Teva | telmisartan | EMEA/H/C/001146 Treatment of essential hypertension in adults |
Withdrawn | yes | no | no | 2010-01-25 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |


