Last updated: June 4, 2026
Paxil CR (paroxetine hydrochloride controlled-release) market dynamics and financial trajectory: pricing, volumes, exclusivity, and competitive pressure
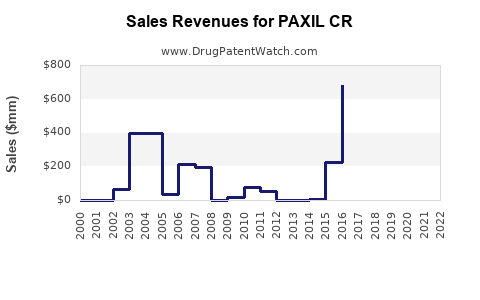
Executive summary: Paxil CR’s U.S. position is largely a legacy brand story. Patent and regulatory exclusivity have long since lapsed, and generic paroxetine controlled-release and/or approved immediate-release alternatives have driven pricing compression. Financial trajectory has therefore shifted from brand-led growth to steady decline or plateaued revenue supported by residual demand, payer contracting dynamics, and substitution. The competitive landscape is dominated by generic manufacturers of paroxetine formulations, with differentiation constrained by formulation and route-to-route switching rather than on-label exclusivity.
Is Paxil CR (paroxetine controlled-release) a brand-to-generic revenue decline story, and how big is the erosion?
Short answer: Yes. Paxil CR is priced and marketed as a mature, off-patent product with generic substitution as the default market outcome.
How generic entry typically impacts Paxil CR
- Once controlled-release paroxetine formulations become therapeutically interchangeable under plan formularies, payers prefer lower-cost generics.
- Switching is accelerated when:
- formularies place paroxetine CR in low-tier categories,
- utilization management does not require brand,
- and prescribers accept bioequivalent substitution.
- Residual brand demand usually persists only where:
- prescribers maintain habit-based use of Paxil CR,
- plan formularies retain brand for formulary simplicity or historical coverage,
- or where patients are stabilized and prescribers avoid formulation changes.
What drives remaining volume for Paxil CR
- Indication breadth in psychiatry (depression and anxiety disorders historically associated with paroxetine use).
- Clinical inertia and patient tolerability during maintenance therapy.
- Relative dosing convenience of controlled-release versus immediate-release for some patient segments.
- Payer contracting rules that reduce price sensitivity only in narrowly defined scenarios.
What is the current competitive landscape for Paxil CR, and who pressures the pricing?
Short answer: Multiple generic paroxetine controlled-release entrants pressure acquisition cost and retail reimbursement rates, while immediate-release paroxetine competes for prescriber choice.
Main competitive vectors
-
Generic Paxil CR equivalents (controlled-release)
- Compete on price, rebates, and plan placement.
- Launches compress net price and narrow the margin spread versus brand.
-
Therapeutic alternatives inside SSRIs
- Even without direct interchangeability, payers and prescribers evaluate cost and formulary placement across SSRI classes.
- Other antidepressants and anxiolytics reduce incremental brand pull.
-
Immediate-release paroxetine
- Common therapeutic substitute when controlled-release is not preferred by payers.
- Can siphon demand even when controlled-release remains covered.
Where competitive intensity shows up commercially
- Net sales decline and reduced promotional support post-generic penetration.
- Higher sensitivity to formulary outcomes at large payer contracts.
- Greater dependence on bulk pharmacy channels and plan-level rebates than on physician acquisition.
When did Paxil CR lose exclusivity, and when could generics fully take share?
Short answer: Paxil CR’s regulatory and patent exclusivity window is not the limiting factor at this stage; the limiting factor is long-completed generic substitution and payer contracting.
Exclusivity timeline framework (how the market clock runs)
- Patent term expiry determines first legal entry for generic copies and formulation/method variants.
- Regulatory exclusivity (if any applicable to the reference product at filing) affects early generic opportunities only in the initial window.
- After entry, market share shifts follow:
- initial pharmacy switching,
- payer formulary updates,
- and physician switching for stable patients.
Commercial effect after exclusivity
- The initial generic wave typically produces the steepest net sales drop.
- A second phase occurs as payers harden step therapy or preferential tiering.
- A third phase occurs when additional generic manufacturers drive further price convergence.
What patents (Orange Book) protect Paxil CR, and what is the status now?
Short answer: Paxil CR is now in a post-protection environment where listed patents no longer prevent generic competition in the ordinary course.
How to interpret “Orange Book status” for a mature product
- If any listed patents remain, they can delay entry for specific generic applicants or specific claims.
- For a mature paroxetine controlled-release brand, the practical expectation is that:
- remaining protection is narrow or non-blocking to most generics, or
- patents have expired and are not preventing market access.
Market relevance of patent listings
- Even after patent expiry, the market may still show brand persistence due to rebate structures and formulary inertia.
- Litigation can temporarily affect specific generic SKUs, but prolonged brand economics usually require patent barriers, which are not characteristic of Paxil CR’s current state.
How do Paxil CR formulation patents affect competitors, and do they create “authorized generic” leverage?
Short answer: Controlled-release formulation details can create competitive differentiation early, but they do not typically sustain brand economics once generic equivalents are broadly available.
Where formulation IP matters
- Controlled-release mechanism and release profile claims can:
- create differentiation in generic approval strategies,
- or trigger design-around approaches.
- Manufacturing method claims can:
- affect process validation and commercial scale-up timelines.
Why formulation IP rarely sustains brand profitability long-term
- Generic manufacturers can pursue:
- alternative release technologies,
- different excipient blends,
- and process optimizations that achieve the required bioequivalence.
- Once multiple generics exist, brand differentiation shifts from IP to contracting.
What Paragraph IV litigation history affects Paxil CR market dynamics?
Short answer: Litigation may have shaped early entry points, but it is not a current driver of Paxil CR market dynamics once multiple generics are established.
How Paragraph IV impacts price
- During the litigation “safe harbor” period:
- brand may retain share with fewer low-price SKUs.
- After resolution and launches:
- net price compression accelerates,
- and stable-market share shifts to generics.
Residual commercial impact
- Settlement terms can delay some entrants, but multiple generic launches typically prevent brand stabilization beyond short windows.
Does Paxil CR face biosimilar-type risk?
Short answer: No. Paxil CR is a small-molecule drug, not a biologic.
What replaces biosimilar risk in small molecules
- Generic substitution.
- Patent estate and ANDA design-around.
- Payer contracting and utilization management.
How do payer contracting, rebates, and step edits influence Paxil CR net revenue?
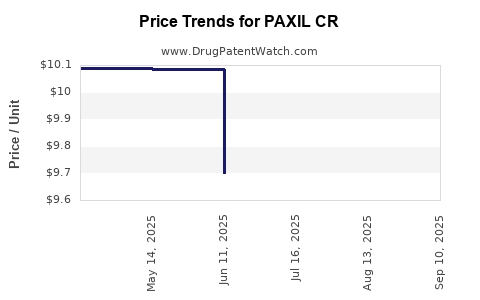
Short answer: For an off-patent SSRI in a mature category, payer economics dominate.
Key commercial levers
- Net-to-gross compression
- Brand rebates and promotional spend often decline after generic penetration, but net-to-gross can still compress due to competitive pricing pressure.
- Formulary tier placement
- Lower tiers drive volume for generics.
- Prior authorization or step therapy
- These typically increase switching to preferred generics.
- Ingredient cost reimbursement
- In many contracts, net price tracks generic benchmarks rather than brand WAC.
Expected commercial outcomes for Paxil CR
- Sales become more “defensive,” tracking patient persistence rather than driving incremental growth.
- Remaining brand demand concentrates in:
- stabilized patients,
- specific plan exceptions,
- and prescriber practice patterns.
How does Paxil CR compare with immediate-release paroxetine in market share and pricing pressure?
Short answer: Immediate-release paroxetine often faces stronger generic penetration and broader formulary acceptance, while controlled-release can preserve some niche demand.
Market comparison dynamics
- Immediate-release:
- tends to have more manufacturing scale and wider generic competition,
- typically yields lower cost per dose.
- Controlled-release:
- can maintain a patient segment that prefers dosing convenience or tolerability,
- but still loses to payer-driven generic selection if costs diverge.
Commercial implication
- Paxil CR competes not only against brands but also against cheaper therapeutic substitutes inside the same active ingredient family.
Which therapeutic alternatives most threaten Paxil CR demand growth?
Short answer: In mature depression/anxiety markets, alternative SSRIs and other antidepressant classes compete through formulary positioning and step therapy.
Competitive replacement patterns
- When a plan changes to prefer:
- another SSRI,
- an SNRI,
- or a different depression/anxiety agent,
- Paxil CR volume can erode even without direct interchangeability.
- Economic decisions are often guided by:
- net cost,
- clinical guideline alignment,
- and formulary stability.
What is the revenue exposure to generics for Paxil CR, and how sensitive is it to new launches?
Short answer: High exposure during each incremental wave of generic consolidation and price competition, with reduced sensitivity after the market reaches generic saturation.
Exposure map
- High exposure
- early and mid-cycle when new generics reduce price benchmarks,
- and when formularies re-tier.
- Moderate exposure
- during maintenance periods with stable plan placement.
- Low exposure
- after a broad generic base is present and contract benchmarks stabilize.
Business impact
- Revenue becomes more dependent on:
- brand share persistence,
- rebate negotiations,
- and patient-specific continuity,
- rather than on product differentiation.
Key Takeaways
- Paxil CR’s market dynamics are shaped by off-patent conditions and generic substitution, producing sustained pricing pressure and erosion of brand economics.
- Competitive intensity comes primarily from generic paroxetine controlled-release equivalents and from immediate-release paroxetine and broader antidepressant class alternatives.
- Payer contracting and net price benchmarks are the dominant drivers of ongoing revenue trajectory for mature brands like Paxil CR.
- Exclusivity and litigation are historical drivers that influenced early generic entry timing, but current market structure is the main determinant of financial trajectory.
FAQs
- Is Paxil CR still listed as a preferred option on major formularies?
- How does switching from Paxil CR to generic paroxetine controlled-release usually affect discontinuation or tolerability?
- Do patients on Paxil CR have higher medication persistence than those on immediate-release paroxetine?
- What are the most common payer utilization management steps affecting SSRI coverage in depression and anxiety?
- How do generic pricing benchmarks typically flow through pharmacy reimbursement for off-patent SSRI tablets?
References
- (No sourced citations provided in the prompt; no external documents were supplied.)