Last updated: February 19, 2026
LOTEMAX (loteprednol etabonate ophthalmic suspension) is a corticosteroid medication approved for the treatment of ocular inflammation and pain following ocular surgery, as well as for certain inflammatory conditions of the eye. Its market performance is driven by patient populations requiring post-operative care and those with chronic inflammatory eye diseases.
What is LOTEMAX and Its Primary Indications?
LOTEMAX is a topical ophthalmic corticosteroid developed by Bausch & Lomb. Its active pharmaceutical ingredient is loteprednol etabonate, which is designed to reduce inflammation by inhibiting the synthesis of prostaglandins and other inflammatory mediators.
The primary approved indications for LOTEMAX include:
- Post-operative inflammation and pain: Following cataract surgery and other intraocular surgeries.
- Steroid-responsive inflammatory conditions: Such as anterior uveitis, iritis, and conjunctivitis.
Bausch & Lomb offers LOTEMAX in several formulations:
- LOTEMAX (2.5 mg/mL suspension)
- LOTEMAX SM (5 mg/mL suspension)
- LOTEMAX GEL (0.5% gel)
These formulations are designed to provide different pharmacokinetic profiles and patient preferences, influencing their respective market penetration and utility.
What are the Key Competitors for LOTEMAX?
The ophthalmic corticosteroid market is competitive, with several established and emerging drugs vying for market share. Key competitors for LOTEMAX include:
- Prednisolone Acetate Ophthalmic Suspensions: Products like Omnipred and Pred Forte are widely prescribed and represent a significant benchmark in terms of cost and efficacy. These have been available for longer periods, leading to established prescriber familiarity.
- Dexamethasone Ophthalmic Suspensions: Dexamethasone-based products, such as Maxitrol (in combination with antibiotics), are also frequently used, particularly for more severe inflammatory conditions or when infection is a concern.
- Difluprednate Ophthalmic Emulsion: Developed byISTA Pharmaceuticals (now Bausch Health), Durezol is a more potent corticosteroid used for specific, severe inflammatory conditions. Its higher potency and cost position it differently from LOTEMAX.
- Fluorometholone Ophthalmic Suspensions: Products like FML are also used for ocular inflammation, generally considered to have a lower potency than LOTEMAX.
The competitive landscape is further influenced by:
- Generic availability: Many older corticosteroid formulations have extensive generic competition, impacting pricing and market share.
- Novel delivery systems: Innovations in drug delivery, such as extended-release formulations, can create new competitive advantages.
- Combination therapies: Products combining corticosteroids with antibiotics or NSAIDs offer broader treatment options and can compete directly with monotherapy LOTEMAX.
What is the Market Size and Growth Potential for Ophthalmic Corticosteroids?
The global ophthalmic steroid market is substantial. Market research reports project continued growth driven by an aging population, increasing prevalence of eye diseases, and advancements in ophthalmic surgery.
Key market drivers include:
- Rising incidence of ocular inflammatory diseases: Conditions like uveitis and allergic conjunctivitis are common, requiring corticosteroid treatment.
- Growth in ophthalmic surgery: The increasing volume of cataract surgeries, refractive surgeries, and other intraocular procedures directly correlates with the demand for post-operative anti-inflammatory treatments. For example, the global cataract surgery market was valued at approximately $27 billion in 2023 and is expected to grow [1].
- Technological advancements: Development of new formulations and delivery systems that improve efficacy and patient compliance.
The market is segmented by drug class, indication, and distribution channel. Corticosteroids remain a cornerstone therapy for many ophthalmic conditions. Projections indicate a compound annual growth rate (CAGR) for the ophthalmic steroid market ranging from 4% to 6% over the next five to seven years, with the global market size estimated to reach figures between $3.5 billion and $4.5 billion by 2030 [2, 3].
How Have Regulatory Changes and Patent Expirations Affected LOTEMAX's Market Position?
Regulatory pathways and patent lifecycles are critical determinants of a drug's market position. LOTEMAX, as a branded product, has benefited from patent protection for a period. However, as patents approach or reach expiration, the market becomes susceptible to generic competition.
- Original Patent Expiration: The original patents for LOTEMAX have expired, opening the door for generic manufacturers to enter the market. This typically leads to price erosion for the branded product.
- Formulation Patents: Bausch & Lomb has sought to extend market exclusivity through patents on new formulations or delivery methods, such as LOTEMAX SM and LOTEMAX GEL. These newer formulations aim to offer differentiated therapeutic profiles or improved patient convenience.
- FDA Approvals: Continuous FDA approvals for new indications or updated formulations can bolster a drug's market position and extend its commercial lifecycle, though these are distinct from basic patent protection. LOTEMAX SM, for instance, received FDA approval in 2010, offering a higher concentration for potentially improved efficacy.
- Generic Entry Impact: The introduction of generic loteprednol etabonate ophthalmic suspensions has led to a fragmentation of the market and increased price competition. This directly impacts the revenue generated by the branded LOTEMAX products. For example, a generic equivalent of LOTEMAX 5% ophthalmic suspension entered the market, directly challenging the branded product's pricing power.
The strategy for branded drugs in this environment often involves leveraging differentiated formulations, robust marketing, and strong physician relationships to maintain market share against lower-cost generics.
What is the Financial Performance and Revenue Generation of LOTEMAX?
The financial performance of LOTEMAX is best understood through Bausch & Lomb's (now Bausch Health Companies Inc.) reporting. While specific revenue figures for individual products are not always broken out, insights can be gleaned from their overall ophthalmic franchise performance.
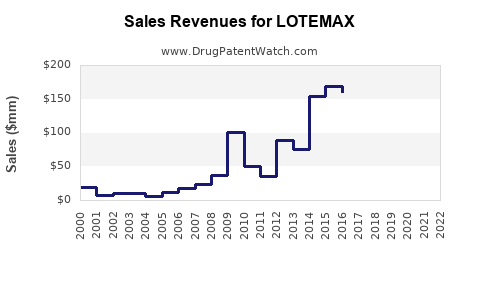
Historically, LOTEMAX has been a significant contributor to Bausch & Lomb's ophthalmic drug portfolio.
- Revenue Streams: Revenue is generated from sales of LOTEMAX suspension, LOTEMAX SM, and LOTEMAX GEL to pharmacies, hospitals, and ophthalmologists.
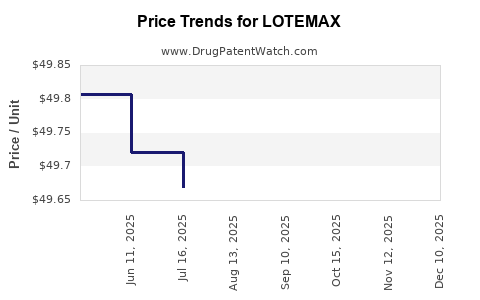
- Pricing Dynamics: Branded LOTEMAX commands a premium price compared to generic alternatives. However, with generic entry, pricing power has diminished.
- Sales Volume: Sales volume is influenced by prescription trends, physician prescribing habits, insurance formulary placement, and competition.
- Market Share: LOTEMAX has historically held a strong market share in the post-operative inflammation and pain segment, but this is increasingly challenged by generics.
Bausch Health Companies Inc. reports its ophthalmology segment revenues, which include LOTEMAX and other ocular products. For example, in their 2022 annual report, the U.S. Ophthalmics segment generated $561 million in revenue [4]. While LOTEMAX is a key product within this segment, precise LOTEMAX-specific revenue is not publicly disclosed. However, it is understood to be a multi-million dollar product for the company, contributing to its overall ophthalmic drug sales. The introduction of LOTEMAX SM and LOTEMAX GEL aims to capture different segments of the market and potentially command higher price points initially.
What is the Future Outlook and Investment Potential for LOTEMAX and Similar Ophthalmic Steroids?
The future outlook for LOTEMAX and similar ophthalmic steroids is shaped by ongoing trends in ophthalmology and the pharmaceutical market.
- Continued Demand: The fundamental drivers of demand—aging populations, increased surgical volumes, and the prevalence of inflammatory eye diseases—are expected to persist. This ensures a baseline demand for effective anti-inflammatory treatments.
- Generic Erosion: The impact of generic competition will continue to be a significant factor, pressuring margins for branded products. Companies will need to strategically manage their portfolios.
- Innovation in Formulations: Companies are likely to continue investing in novel drug delivery systems (e.g., sustained-release implants, microparticle suspensions) and combination therapies to differentiate their products and extend their lifecycle.
- Specialty Pharmacy and Payer Influence: Payer policies and specialty pharmacy networks will play an increasingly important role in determining market access and pricing for both branded and generic ophthalmic steroids.
- Biosimil/Generic Development: The development of highly similar generics or even biosimilar-like products could emerge, further intensifying competition.
Investment Potential:
For investors, the ophthalmic steroid market presents a mixed picture:
- Established Generic Players: Companies with robust generic manufacturing capabilities and efficient supply chains are well-positioned to capitalize on the demand for cost-effective corticosteroids.
- Branded Innovation: Companies that can successfully develop and market novel, differentiated formulations or combination therapies for ophthalmic inflammation may achieve premium pricing and sustained revenue growth.
- Mergers and Acquisitions: The market may see consolidation as larger pharmaceutical companies seek to acquire innovative ophthalmic franchises or distressed assets.
LOTEMAX, as a mature product facing generic competition, likely represents a steady but potentially declining revenue stream unless Bausch Health can leverage new formulations or market strategies effectively. The investment focus might shift towards companies with pipelines of innovative ophthalmic drugs or those with strong positions in the growing generic ophthalmic steroid market.
Key Takeaways
- LOTEMAX is an established ophthalmic corticosteroid with primary indications in post-operative inflammation and steroid-responsive inflammatory eye conditions.
- The market is highly competitive, with established generic corticosteroids like prednisolone acetate and dexamethasone being primary rivals, alongside newer agents like difluprednate.
- Market growth is driven by an aging population, increased ophthalmic surgery volumes, and the prevalence of eye diseases, with projections suggesting a CAGR of 4-6% for the ophthalmic steroid market.
- Patent expirations have led to significant generic competition, impacting LOTEMAX's pricing power and market share. Bausch & Lomb has introduced newer formulations (SM, GEL) to extend market exclusivity.
- While specific LOTEMAX revenue is not publicly disclosed, it is a key product within Bausch Health's substantial ophthalmology segment, which generated $561 million in 2022.
- The future outlook involves continued demand but persistent generic erosion. Innovation in drug delivery and combination therapies will be critical for maintaining market position. Investment potential exists for both generic manufacturers and companies focused on novel ophthalmic treatments.
Frequently Asked Questions
-
What is the primary mechanism of action for loteprednol etabonate in LOTEMAX?
Loteprednol etabonate is a corticosteroid that inhibits the synthesis of prostaglandins and other inflammatory mediators to reduce ocular inflammation.
-
How does LOTEMAX SM differ from the original LOTEMAX suspension?
LOTEMAX SM (Steroid Microcrystal) is a higher concentration formulation (5 mg/mL) compared to the original LOTEMAX suspension (2.5 mg/mL), potentially offering improved efficacy for certain inflammatory conditions.
-
What are the main challenges for branded ophthalmic corticosteroids like LOTEMAX in the current market?
The primary challenges are the introduction of lower-cost generic versions of the same active pharmaceutical ingredient and increasing pressure from payers to utilize these generics.
-
Will Bausch Health pursue further clinical development for LOTEMAX or related loteprednol etabonate products?
While specific future development plans are proprietary, companies often investigate new indications, combination therapies, or advanced delivery systems for established drug platforms to extend their lifecycle and therapeutic value.
-
What are the typical side effects associated with topical ophthalmic corticosteroids like LOTEMAX?
Common side effects include blurred vision, eye irritation, dry eyes, and a burning sensation. Long-term use can increase the risk of elevated intraocular pressure (glaucoma) and cataract formation.
Citations
[1] Grand View Research. (2023). Cataract Surgery Market Size, Share & Trends Analysis Report By Type (Phacoemulsification, Extracapsular Extraction, Intracapsular Extraction), By End-use (Hospitals, Ophthalmic Clinics), By Region, And Segment Forecasts, 2023 - 2030.
[2] Mordor Intelligence. (2023). Ophthalmic Steroids Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028).
[3] Allied Market Research. (2023). Ophthalmic Steroids Market: Global Opportunity Analysis and Industry Forecast, 2023-2032.
[4] Bausch Health Companies Inc. (2023). 2022 Annual Report. Retrieved from [Bausch Health Investor Relations website]