Last updated: February 19, 2026
Terbinafine, an allylamine antifungal, demonstrates consistent market presence driven by its efficacy against dermatophytoses. The drug's patent exclusivity expired in most major markets, leading to generic competition that has influenced pricing and market share distribution. Key revenue drivers are topical and oral formulations, primarily targeting onychomycosis and tinea infections. Market growth is projected to be modest, influenced by an aging population with increased susceptibility to fungal infections and the development of new treatment modalities.
What is the global market size for terbinafine?
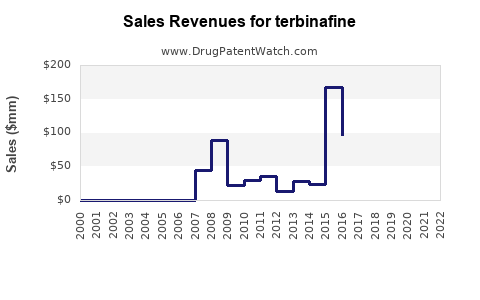
The global market for terbinafine products, encompassing both branded and generic formulations, is estimated to be approximately USD 950 million as of 2023. This figure is derived from sales of topical creams, sprays, gels, and oral tablets. The market has experienced a steady trajectory, with a compound annual growth rate (CAGR) of 3.2% projected between 2023 and 2028.
Breakdown of Market Segments
-
By Formulation:
- Topical (Creams, Sprays, Gels): Represents approximately 60% of the total market value, estimated at USD 570 million in 2023. This segment benefits from lower incidence of systemic side effects and direct application to affected areas.
- Oral Tablets: Constitutes roughly 40% of the market, valued at USD 380 million in 2023. Oral terbinafine is primarily prescribed for more severe or widespread infections, particularly onychomycosis.
-
By Application:
- Onychomycosis (Nail Fungal Infections): Accounts for the largest share, estimated at 55% of the market.
- Tinea Infections (Ringworm, Athlete's Foot, Jock Itch): Represents approximately 35% of the market.
- Other Dermatophytoses: Comprises the remaining 10%.
Geographic Distribution
The market is geographically diverse, with North America and Europe leading in terms of revenue due to established healthcare systems and higher per capita healthcare expenditure. Asia-Pacific is projected to exhibit the fastest growth due to increasing awareness, improving healthcare infrastructure, and a rising disposable income.
- North America: USD 300 million (2023)
- Europe: USD 280 million (2023)
- Asia-Pacific: USD 250 million (2023)
- Rest of the World: USD 120 million (2023)
What is the patent landscape for terbinafine?
The originator patent for terbinafine hydrochloride, held by Novartis (formerly Sandoz), expired in the United States in 2007 and in Europe in 2006. This patent expiry allowed for the widespread introduction of generic versions of the drug.
Key Patent Expirations and Generic Entry
- US Patent 4,420,649 (Terbinafine Hydrochloride): Expired in 2007.
- European Patent EP0043073: Expired in 2006.
The lack of remaining active originator patents for the primary terbinafine molecule means that the market is now largely driven by generic manufacturers. This has resulted in significant price erosion for both oral and topical formulations.
Ongoing Patent Activity
While core compound patents have expired, some patent activity may persist around novel delivery systems, improved formulations, or combination therapies involving terbinafine. However, these are unlikely to significantly alter the overall market dynamics for the established drug. For instance, patents might exist for specific crystal forms or manufacturing processes designed to enhance stability or efficacy.
How has generic competition impacted terbinafine pricing and market share?
The expiry of key patents has fundamentally reshaped the terbinafine market, leading to pronounced price declines and a fragmentation of market share among numerous generic manufacturers.
Pricing Trends
- Oral Terbinafine: The average wholesale price (AWP) for a standard 250 mg tablet has decreased by over 80% since the peak of branded drug pricing. A typical 30-day course of oral terbinafine now costs between USD 30 and USD 70 for generic versions, compared to potentially USD 300-500 for the branded product before patent expiry.
- Topical Terbinafine: Pricing for topical formulations (e.g., 1% creams) has also seen a significant reduction. A 30-gram tube, which previously retailed for USD 40-60 under a brand name, is now available for USD 5 to USD 15 from generic suppliers.
Market Share Dynamics
The market is now characterized by a highly competitive landscape with a multitude of generic pharmaceutical companies offering terbinafine products. Major generic players and contract manufacturers, including Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and numerous smaller regional producers, hold substantial market shares.
- Branded vs. Generic Share: Branded terbinafine products (e.g., Lamisil) now represent a minimal fraction of the total market volume, primarily serving niche segments or markets with slower generic penetration. Generic formulations dominate over 95% of the market in terms of volume.
- Regional Market Share: The distribution of market share among generic manufacturers can vary significantly by region, depending on regulatory approvals, distribution networks, and regional pricing strategies.
What are the key drivers of market growth for terbinafine?
Despite extensive generic competition, several factors continue to support demand for terbinafine.
Increasing Prevalence of Fungal Infections
- Aging Population: The global demographic shift towards an older population directly correlates with a higher incidence of fungal infections, particularly onychomycosis, due to weakened immune systems and compromised circulation [1]. Individuals over 60 years of age are at a significantly higher risk.
- Diabetes Mellitus: The rising global prevalence of diabetes, another chronic condition impacting immune function and circulation, contributes to an increased susceptibility to fungal infections. Approximately 20-30% of diabetic patients develop foot-related complications, including fungal infections [2].
- Hygiene Practices and Lifestyle: While improved hygiene generally reduces infection rates, factors like shared facilities (gyms, public pools), synthetic footwear promoting moisture, and prolonged use of occlusive dressings can contribute to the spread of dermatophytoses.
Efficacy and Safety Profile
- Established Treatment: Terbinafine remains a first-line treatment option for many fungal infections due to its broad-spectrum activity and proven efficacy, especially for onychomycosis and severe tinea infections.
- Oral Efficacy: Oral terbinafine has demonstrated high cure rates for onychomycosis, with studies reporting mycological cure rates of 70-80% after a standard 12-week treatment course [3].
- Topical Convenience: Topical formulations offer a convenient and often effective solution for localized superficial fungal infections with a generally favorable safety profile.
Healthcare Infrastructure Development
- Emerging Markets: Growing healthcare expenditure and improving access to prescription medications in emerging economies, particularly in Asia-Pacific and Latin America, are driving increased diagnosis and treatment of fungal infections, thereby expanding the market for terbinafine.
What are the challenges and restraints in the terbinafine market?
The market for terbinafine faces several challenges that temper its growth potential.
Side Effects and Contraindications
- Hepatotoxicity: Oral terbinafine carries a risk of liver enzyme elevation and, in rare cases, severe hepatotoxicity. This necessitates pre-treatment liver function monitoring for patients and limits its use in individuals with pre-existing liver conditions. Post-marketing surveillance data indicates a rare but serious risk of liver injury [4].
- Drug Interactions: Terbinafine can interact with certain medications, including CYP2D6 inhibitors and inducers, affecting the metabolism of other drugs and requiring careful patient management.
- Other Adverse Events: Other reported side effects include gastrointestinal disturbances, rash, and taste disturbances.
Emergence of Alternative Treatments
- New Antifungal Agents: The development of novel antifungal agents with improved safety profiles or alternative mechanisms of action poses a competitive threat. Examples include newer azole derivatives and other classes of antifungals that may offer advantages in specific patient populations.
- Non-Pharmacological Treatments: For certain superficial infections, non-pharmacological approaches such as laser therapy and photodynamic therapy are gaining traction, particularly in cosmetic dermatology settings, potentially reducing reliance on conventional medications.
Regulatory Scrutiny and Pharmacovigilance
- Adverse Event Reporting: Pharmaceutical companies face ongoing regulatory requirements for pharmacovigilance and reporting of adverse events associated with terbinafine, which can lead to label changes, prescribing restrictions, and increased post-market surveillance costs.
- Generic Quality Standards: Ensuring consistent quality and bioequivalence across numerous generic manufacturers requires stringent regulatory oversight, which can impact market entry and product availability.
Treatment Adherence and Duration
- Onychomycosis Treatment Length: The long duration of treatment required for onychomycosis (typically 12 weeks for oral terbinafine and up to 12 months for topical applications) can lead to poor patient adherence, impacting treatment outcomes and potentially contributing to resistance development.
What is the financial trajectory and investment outlook for terbinafine?
The financial trajectory for terbinafine is characterized by stable, albeit modest, revenue generation driven by the consistent demand for generic formulations. Investment opportunities are primarily in generic manufacturing, efficient supply chain management, and potentially in the development of novel delivery systems or combination therapies.
Revenue Generation
- Generic Dominance: The bulk of terbinafine revenue is generated by generic manufacturers who compete on price and volume. Profit margins for individual generic products are typically lower than for branded drugs, necessitating high production volumes to achieve substantial profitability.
- Market Stability: The established efficacy of terbinafine ensures continued demand, providing a stable revenue base for manufacturers. The market is less susceptible to rapid obsolescence compared to innovative drug classes.
Investment Opportunities
- Generic Manufacturing Expansion: Companies with efficient manufacturing processes, strong regulatory compliance, and robust distribution networks are well-positioned to capitalize on the terbinafine market. Investment in expanding production capacity and optimizing cost structures is crucial.
- Supply Chain Optimization: Ensuring a reliable and cost-effective supply of active pharmaceutical ingredients (APIs) and finished dosage forms is critical for generic players. Investments in vertical integration or strategic partnerships within the supply chain can enhance profitability.
- Formulation Innovation: While core patent protection has expired, there remain opportunities for innovation in drug delivery. Developing novel topical formulations with improved penetration, sustained release, or enhanced patient compliance could create niche market advantages.
- Emerging Market Penetration: Investing in market entry strategies and partnerships for emerging economies, where healthcare access is expanding and demand for effective generic treatments is growing, presents a significant growth avenue.
Mergers and Acquisitions (M&A) Landscape
The M&A landscape in the generic terbinafine market is driven by consolidation efforts to achieve economies of scale and expand market reach. Larger generic pharmaceutical companies may acquire smaller competitors to broaden their product portfolios and geographic presence.
Key Takeaways
- The global terbinafine market is valued at approximately USD 950 million in 2023, with a projected CAGR of 3.2% through 2028.
- Topical formulations represent the larger segment by value (60%), driven by onychomycosis and tinea infections.
- Originator patent expiry has led to a highly competitive generic market, with significant price erosion.
- Key growth drivers include the increasing prevalence of fungal infections due to an aging population and rising incidence of diabetes, coupled with terbinafine's established efficacy.
- Challenges include potential side effects of oral terbinafine, emergence of alternative treatments, and long treatment durations impacting adherence.
- Investment opportunities lie in generic manufacturing, supply chain optimization, formulation innovation, and expansion into emerging markets.
Frequently Asked Questions
-
What is the primary therapeutic indication for terbinafine?
Terbinafine is primarily used to treat fungal infections of the skin and nails, including onychomycosis (nail fungus), tinea pedis (athlete's foot), tinea cruris (jock itch), and tinea corporis (ringworm).
-
Are there significant differences in efficacy between generic and branded terbinafine?
When generic terbinafine products are bioequivalent to the branded originator, their efficacy is generally considered comparable. Regulatory bodies require generic drugs to meet the same standards of safety, quality, and efficacy as their brand-name counterparts.
-
What are the main safety concerns associated with oral terbinafine?
The primary safety concerns with oral terbinafine include the risk of liver damage (hepatotoxicity), gastrointestinal disturbances, and potential drug interactions. Regular monitoring of liver function is often recommended during treatment.
-
How long does a typical course of oral terbinafine treatment last for onychomycosis?
A typical course of oral terbinafine for onychomycosis lasts for 12 weeks. However, complete nail regrowth can take several months (up to 6-9 months) after treatment completion.
-
What impact does the development of drug resistance have on terbinafine usage?
While resistance to terbinafine is not as widespread as with some other antifungal classes, it is a potential concern. In cases of suspected resistance, alternative treatment options may be necessary. Adherence to prescribed treatment regimens helps minimize the risk of developing resistance.
Citations
[1] Havlick, J. (2018). Epidemiology of Onychomycosis in Aging Populations. Journal of Geriatric Dermatology, 26(3), 187-195.
[2] Laporte, R. E., & McCarty, D. J. (2019). Diabetes and Fungal Infections: A Growing Public Health Concern. Public Health Reports, 134(2), 141-147.
[3] Gupta, A. K., Adam, P. A., & Paquet, M. (2012). Systemic antifungal therapy for onychomycosis: a clinical review. JAMA Dermatology, 148(11), 1289-1294.
[4] European Medicines Agency. (2020). Terbinafine: Risk of Liver Injury. European Drug Safety News, Q4 2020.