Salix Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SALIX PHARMS, and what generic alternatives to SALIX PHARMS drugs are available?
SALIX PHARMS has nine approved drugs.
There are forty US patents protecting SALIX PHARMS drugs.
There are three hundred and twenty-two patent family members on SALIX PHARMS drugs in forty-three countries and eighty-six supplementary protection certificates in eleven countries.
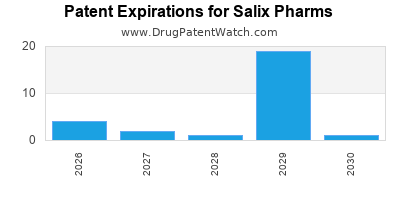
Drugs and US Patents for Salix Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-002 | Mar 24, 2010 | RX | Yes | Yes | 7,902,206 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Salix Pharms | METOZOLV ODT | metoclopramide hydrochloride | TABLET, ORALLY DISINTEGRATING;ORAL | 022246-002 | Sep 4, 2009 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | RX | Yes | Yes | 9,492,445 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-001 | Apr 24, 2008 | RX | Yes | Yes | 8,420,663 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | RX | Yes | Yes | 9,180,125 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-001 | May 25, 2004 | RX | Yes | Yes | 9,271,968 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Salix Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Salix Pharms | METOZOLV ODT | metoclopramide hydrochloride | TABLET, ORALLY DISINTEGRATING;ORAL | 022246-001 | Sep 4, 2009 | 6,413,549 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 6,559,158 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | 10,376,584 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | 8,552,025 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 8,552,025 | ⤷ Try a Trial |
| Salix Pharms | OSMOPREP | sodium phosphate, dibasic, anhydrous; sodium phosphate, monobasic, monohydrate | TABLET;ORAL | 021892-001 | Mar 16, 2006 | 5,616,346 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for SALIX PHARMS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 12 mg/0.6 mL | ➤ Subscribe | 2015-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2016-09-06 |
| ➤ Subscribe | Tablets | 1.102 g and 0.398 g | ➤ Subscribe | 2008-04-09 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2019-01-28 |
| ➤ Subscribe | For Oral Solution | 100 g, 7.5 g, 2.691 g, 1.015 g, 5.9 g and 4.7 g per pouch | ➤ Subscribe | 2007-11-27 |
| ➤ Subscribe | Injection | 8 mg/0.4 mL | ➤ Subscribe | 2015-09-08 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2010-08-24 |
| ➤ Subscribe | Tablets | 550 mg | ➤ Subscribe | 2015-12-18 |
International Patents for Salix Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 5737825 | ⤷ Try a Trial |
| Taiwan | I285107 | ⤷ Try a Trial |
| Mexico | 2018015087 | ⤷ Try a Trial |
| European Patent Office | 2208730 | ⤷ Try a Trial |
| South Korea | 20110098015 | ⤷ Try a Trial |
| Poland | 3608322 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Salix Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1666481 | 17C1031 | France | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB,EVENTUELLEMENT SOUS LA FORME D'UN SEL PHARMACEUTIQUEMENT ACCEPTABLE,DONT LE CITRATE DE SODIUM; REGISTRATION NO/DATE: EU/1/17/1178 20170324 |
| 2465580 | SPC/GB21/030 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: CABOTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF, INCLUDING CABOTEGRAVIR SODIUM.; REGISTERED: UK EU/1/20/1481 (NI) 20201221; UK PLGB 35728/0055-57 20201221 |
| 0454511 | SPC/GB99/008 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: 2-N-BUTYL-4-SPIROCYCLOPENTANE-1-((2'-(TETRAZOL-5-YL)BIPHENYL-4-YL)METHYL)-2-IMIDAZOLIN-5-ONE)(GENERIC NAME IRBESARTAN) OPTIONALLY IN THE FORM OF ONE OF ITS SALTS AND HYDROCHLOROTHIAZIDE; REGISTERED: UK EU/1/98/086/001 19981015; UK EU/1/98/086/002 19981015; UK EU/1/98/086/003 19981015; UK EU/1/98/086/004 19981015; UK EU/1/98/086/005 19981015; UK EU/1/98/086/006 19981015 |
| 2203431 | CA 2015 00014 | Denmark | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REG. NO/DATE: EU/1/14/983 20150115 |
| 0502314 | SPC/GB02/037 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: TELMISARTAN, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND HYDROCHLOROTHIAZIDE; REGISTERED: UK EU/1/02/213/001 20020419; UK EU/1/02/213/002 20020419; UK EU/1/02/213/003 20020419; UK EU/1/02/214/004 20020419; UK EU/1/02/213/005 20020419; UK EU/1/02/213/006 20020419; UK EU/1/02/213/007 20020419; UK EU/1/02/213/008 20020419; UK EU/1/02/213/009 20020419; UK EU/1/02/213/010 20020419 |
| 0290047 | 97C0108 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MANGAFODIPIR TRISODIUM (ANHYDROUS) CORRESP. MANGAFODIPIR; REGISTRATION NO/DATE: EU/1/97/040/001 19970522 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.