Salix Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SALIX PHARMS, and when can generic versions of SALIX PHARMS drugs launch?
SALIX PHARMS has nine approved drugs.
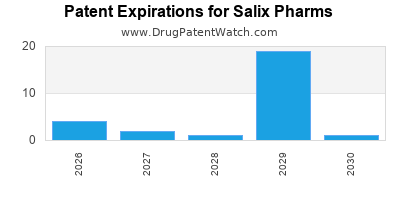
There are forty US patents protecting SALIX PHARMS drugs.
There are three hundred and twenty-two patent family members on SALIX PHARMS drugs in forty-three countries and eighty-six supplementary protection certificates in eleven countries.
Drugs and US Patents for Salix Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Salix Pharms | VISICOL | sodium phosphate, dibasic, anhydrous; sodium phosphate, monobasic, monohydrate | TABLET;ORAL | 021097-001 | Sep 21, 2000 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-002 | Mar 24, 2010 | RX | Yes | Yes | 10,456,384 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | RX | Yes | Yes | 10,376,584 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-001 | May 25, 2004 | RX | Yes | Yes | 8,518,949 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-001 | Apr 24, 2008 | RX | Yes | Yes | 8,822,490 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Salix Pharms | XIFAXAN | rifaximin | TABLET;ORAL | 021361-001 | May 25, 2004 | RX | Yes | Yes | 8,193,196 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Salix Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 6,559,158 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 8,552,025 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-002 | Sep 27, 2010 | 9,669,096 | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | 9,669,096 | ⤷ Try a Trial |
| Salix Pharms | PEPCID | famotidine | FOR SUSPENSION;ORAL | 019527-001 | Feb 2, 1987 | 4,283,408*PED | ⤷ Try a Trial |
| Salix Pharms | RELISTOR | methylnaltrexone bromide | SOLUTION;SUBCUTANEOUS | 021964-003 | Sep 27, 2010 | 8,552,025 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for SALIX PHARMS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 1.102 g and 0.398 g | ➤ Subscribe | 2008-04-09 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2019-01-28 |
| ➤ Subscribe | For Oral Solution | 100 g, 7.5 g, 2.691 g, 1.015 g, 5.9 g and 4.7 g per pouch | ➤ Subscribe | 2007-11-27 |
| ➤ Subscribe | Injection | 8 mg/0.4 mL | ➤ Subscribe | 2015-09-08 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2010-08-24 |
| ➤ Subscribe | Tablets | 550 mg | ➤ Subscribe | 2015-12-18 |
| ➤ Subscribe | Injection | 12 mg/0.6 mL | ➤ Subscribe | 2015-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2016-09-06 |
International Patents for Salix Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2007512336 | ⤷ Try a Trial |
| Portugal | 1676848 | ⤷ Try a Trial |
| South Korea | 20120025578 | ⤷ Try a Trial |
| Canada | 2672427 | ⤷ Try a Trial |
| Australia | 2012216409 | ⤷ Try a Trial |
| South Korea | 100855084 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Salix Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0480717 | SPC/GB98/025 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MONTELUKAST, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, PREFERABLY MONTELUKAST SODIUM; REGISTERED: FI 12766 19970825; FI 12767 19970825; UK 00025/0357 19980115; UK 00025/0358 19980115 |
| 2932970 | SPC/GB18/041 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION COMPRISING DOLUTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF (E.G. DOLUTEGRAVIR SODIUM) AND RILPIVIRINE OR A PHARAMACEUTICALLY ACCEPTABLE SALT THEREOF (E.G. RILPIVIRINE HYDROCHLORIDE); REGISTERED: UK EU/1/18/1282 20180518; UK PLGB 35728/0055 20180518; UK PLGB 35728/0056 20180518; UK PLGB 35728/0057 20180518 |
| 1948158 | 93075 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL ET VALSARTAN, SOUS FORME DE COMPLEXE SODIQUE SACUBITRIL VALSARTAN, C'EST-A-DIRE DE (3-((1S,3R)-1-BIPHENYL-4-YLMETHYL-3-ETHOXYCARBONYL-1-BUTYLCARBAMOYL)PROPIONATE-(S)-3'-METHYL-2-(PENTANYOL(2''-(TETRAZOL-5-YLATE)BIPHENYL-4'-YLMETHYL)AMINO)BUTYRATE) DE TRISODIUM HEMIPENTAHYDRATE; AUTHORISATION NUMBER AND DATE: EU/1/15/1058 20151123 |
| 0579826 | SPC/GB02/042 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ERTAPENEM ((1R,5S,6S,8R,2'S,4'S)-2-(2-(3-CARBOXYPHENYLCARBAMOYL)PYRROLIDIN-4-YLTHIO)-6-(1-HYDROXYETHYL)-1-METHYLCARBAPENEM-3-CARBOXYLIC ACID), A PHARMACEUTICALLY ACCEPTABLE SALT OR IN VIVO HYDROLYSABLE ESTER THEREOF, ESPECIALLY AS THE MONOSODIUM SALT ERTA; REGISTERED: UK EU/1/02/216/001 20020422 |
| 2203431 | 1590018-6 | Sweden | ⤷ Try a Trial | PRODUCT NAME: DASABUVIR OR A SALT THEREOF, INCLUDING DASABUVIR SODIUM MONOHYDRATE; REG. NO/DATE: EU/1/14/983 20150119 |
| 0503785 | 91330 | Luxembourg | ⤷ Try a Trial | CERTIFICATE TITLE: UNE COMBINAISON D'OLMESARTAN MEDOXOMIL, OPTIONNELLEMENT SOUS LA FORME D'UN SEL PHARMACEUTIQUEMENT ACCEPTABLE ET D'HYDROCHLOROTHIAZIDE (OLMETEC PLUS) |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.