Last updated: March 27, 2026
Janssen, the pharmaceutical arm of Johnson & Johnson, maintains a significant presence across multiple therapeutic areas, including immunology, oncology, neuroscience, infectious diseases, and vaccines. This analysis assesses Janssen’s market position, core strengths, and strategic initiatives shaping its competitive landscape.
What is Janssen’s Market Position?
Janssen ranks among the top global biopharmaceutical companies based on revenue, portfolio breadth, and R&D investment. It generated approximately $53 billion in 2022, with a focus on innovative medicines and vaccines. Its core markets include the United States, Europe, China, and emerging economies.
Market Share Overview
| Therapeutic Area |
Estimated Global Market Share (2022) |
Key Products |
Notable Competitors |
| Oncology |
12% |
Imbruvica, Darzalex, Relprevv |
Roche, Novartis, Merck |
| Immunology |
15% |
Stelara, Tremfya |
AbbVie, Eli Lilly |
| Neuroscience |
8% |
Invega, Risperdal |
Novartis, Lilly |
| Infectious Diseases |
10% |
Johnson & Johnson vaccines |
Moderna, Pfizer |
| Vaccines |
9% |
Johnson & Johnson COVID vaccine |
Pfizer, Moderna |
These figures position Janssen as a leading innovator with a strong foothold, especially in immunology and oncology.
What Are Janssen’s Key Strengths?
Broad and Innovative Portfolio
Janssen invests approximately $9 billion annually in R&D. Its pipeline includes over 100 investigational drugs, with a focus on biologics and gene therapies. Notable approved products include Imbruvica (mantle cell lymphoma), Stelara (psoriasis), and the COVID-19 vaccine Ad26.COV2.S.
Focus on Precision Medicine
Janssen emphasizes targeted therapies, leveraging biomarker-driven development. This approach increases the likelihood of regulatory approval and market adoption.
Robust Global Infrastructure
The company operates manufacturing facilities in over 30 countries. Its supply chain resilience supports rapid scale-up during health crises, exemplified by vaccine production during the COVID-19 pandemic.
Strategic Collaborations and Acquisitions
Janssen collaborates with biotech firms and academic institutions to access cutting-edge technologies. Recent acquisitions include Momenta Pharmaceuticals (immunology and biosimilars, 2021) and a licensing agreement with BeiGene for BTK inhibitors.
What Strategic Initiatives Does Janssen Pursue?
Focused Innovation in High-Growth Areas
Janssen prioritizes oncology, immunology, and vaccines. It launched multiple initiatives to expand its pipeline, including gene therapies for hemophilia and rare diseases.
Digital Transformation and Data Analytics
Janssen invests in AI and data analytics to streamline R&D and personalized medicine. Digital platforms facilitate patient engagement and real-world evidence collection.
Expansion in Emerging Markets
Targeting high-growth regions, Janssen invests in local manufacturing and clinical trials. It aims for a 20% revenue increase in China over the next five years.
Sustainability and Access Strategies
Janssen emphasizes affordable access programs and sustainable manufacturing practices, aligning with Johnson & Johnson's global commitments.
How Does Janssen Compare to Competitors?
| Company |
Estimated 2022 Revenue |
R&D Investment (2022) |
Key Differentiators |
| Pfizer |
$100 billion |
$13.8 billion |
Broad portfolio, mRNA vaccines |
| Roche |
$63 billion |
$11 billion |
Diagnostics, personalized oncology |
| Novartis |
$51 billion |
$9.8 billion |
Gene therapies, biosimilars |
| Johnson & Johnson |
$94 billion |
$12.7 billion |
Consumer health, vaccines, pipeline |
Janssen maintains a competitive edge through targeted innovation, fewer patent cliffs than peers, and a focus on high-value biologics.
Key Challenges and Risks
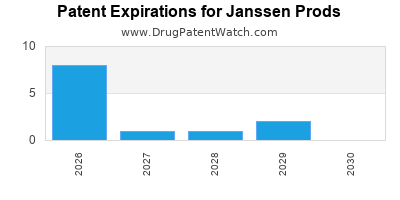
- Patent expirations threaten revenue streams in mature markets by 2025.
- Pricing pressures due to healthcare cost containment policies.
- Regulatory hurdles in emerging markets.
- Competition from biosimilars and generics.
Strategic Recommendations
- Accelerate pipeline development in gene therapy and personalized medicine.
- Strengthen digital patient engagement platforms.
- Expand collaborations with biotech startups.
- Focus on sustainable manufacturing to meet ESG requirements.
- Invest in market access initiatives in emerging economies.
Key Takeaways
Janssen sustains a position as a top-tier pharmaceutical innovator with deep pipeline assets, globally integrated operations, and a focus on high-growth therapeutic areas. Its strategic investments in digital health, emerging markets, and targeted biologics support growth resilience amid patent expiries and competitive pressures.
FAQs
-
How does Janssen's pipeline compare to competitors?
Janssen has over 100 pipeline candidates, emphasizing biologics and gene therapies. Its pipeline is considered robust, especially in oncology and immunology, with promising early-stage assets.
-
What are Janssen’s main growth areas?
Oncology, immunology, and vaccines represent primary growth targets. Recent launches and pipeline progress support an optimistic outlook.
-
How is Janssen addressing patent expiries?
Through pipeline expansion, biosimilar development, and strategic acquisitions, Janssen seeks to mitigate revenue loss from patent cliffs.
-
What role does digital transformation play for Janssen?
It enhances R&D efficiency, facilitates patient engagement, and improves real-world evidence collection, enabling more precise therapies.
-
Where is Janssen expanding in emerging markets?
The company targets China, India, and Latin America, investing in manufacturing and clinical infrastructure to tap into local demand and reduce access barriers.
References
[1] Johnson & Johnson. (2022). Annual Report. https://johnsonandjohnson.com/investor-relations/financial-reporting
[2] EvaluatePharma. (2023). World Preview 2022, outlook to 2027. https://evaluate.com
[3] IQVIA. (2022). The Global Use of Medicine in 2022. https://iqvia.com
[4] IBISWorld. (2022). Pharmaceutical Industry in the US. https://ibisworld.com