Emd Serono Inc Company Profile
✉ Email this page to a colleague
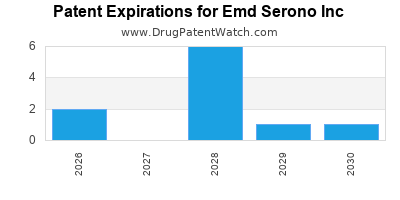
What is the competitive landscape for EMD SERONO INC, and when can generic versions of EMD SERONO INC drugs launch?
EMD SERONO INC has six approved drugs.
There are thirteen US patents protecting EMD SERONO INC drugs.
There are one hundred and ninety-four patent family members on EMD SERONO INC drugs in forty-two countries and eighty-eight supplementary protection certificates in sixteen countries.
Summary for Emd Serono Inc
| International Patents: | 194 |
| US Patents: | 13 |
| Tradenames: | 6 |
| Ingredients: | 5 |
| NDAs: | 6 |
| Drug Master File Entries: | 2 |
Drugs and US Patents for Emd Serono Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Emd Serono Inc | GLUCOPHAGE | metformin hydrochloride | TABLET;ORAL | 020357-001 | Mar 3, 1995 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Emd Serono Inc | TEPMETKO | tepotinib hydrochloride | TABLET;ORAL | 214096-001 | Feb 3, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-001 | Sep 26, 1997 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Emd Serono Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-002 | Sep 26, 1997 | 4,517,181 | ⤷ Try a Trial |
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-002 | Sep 26, 1997 | 4,703,035 | ⤷ Try a Trial |
| Emd Serono Inc | GLUCOPHAGE XR | metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 021202-004 | Apr 11, 2003 | 6,660,300 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Emd Serono Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Argentina | 066543 | ⤷ Try a Trial |
| South Korea | 20060015487 | ⤷ Try a Trial |
| Israel | 274777 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Emd Serono Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1412357 | 77 5006-2008 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: SITAGLIPTIN A METFORMIN; NAT. REG.NO/DATE: EU/1/08/455/001-014, EU/1/08/456/001-014, EU/1/08/457/001-014 20080716; FIRST REG.:CH 58450 01-03 20080408 |
| 1827461 | 122018000021 | Germany | ⤷ Try a Trial | PRODUCT NAME: CLADRIBIN; REGISTRATION NO/DATE: EU/1/17/1212 20170822 |
| 2498758 | 132020000000034 | Italy | ⤷ Try a Trial | PRODUCT NAME: METFORMINA O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE; SAXAGLIPTIN O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE; DAPAGLIFLOZIN O UN SUO SOLVATO FARMACEUTICAMENTE ACCETTABILE.(QTRILMET); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/19/1401, 20191113 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.