Bristol Myers Squibb Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BRISTOL MYERS SQUIBB, and what generic alternatives to BRISTOL MYERS SQUIBB drugs are available?
BRISTOL MYERS SQUIBB has seventy approved drugs.
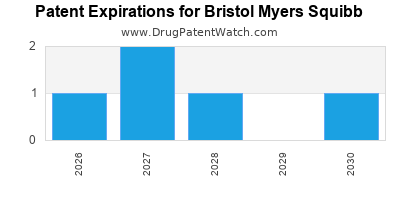
There are thirteen US patents protecting BRISTOL MYERS SQUIBB drugs.
There are five hundred and ninety-six patent family members on BRISTOL MYERS SQUIBB drugs in fifty-one countries and one hundred and forty-three supplementary protection certificates in eighteen countries.
Summary for Bristol Myers Squibb
| International Patents: | 596 |
| US Patents: | 13 |
| Tradenames: | 60 |
| Ingredients: | 55 |
| NDAs: | 70 |
Drugs and US Patents for Bristol Myers Squibb
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | BUSPAR | buspirone hydrochloride | TABLET;ORAL | 018731-003 | Apr 22, 1996 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | IDHIFA | enasidenib mesylate | TABLET;ORAL | 209606-001 | Aug 1, 2017 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | VIDEX | didanosine | TABLET, CHEWABLE;ORAL | 020154-005 | Oct 9, 1991 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | COUMADIN | warfarin sodium | TABLET;ORAL | 009218-018 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | COUMADIN | warfarin sodium | TABLET;ORAL | 009218-023 | Aug 24, 1993 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bristol Myers Squibb | AMNESTROGEN | estrogens, esterified | TABLET;ORAL | 083266-004 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bristol Myers Squibb
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | VIDEX | didanosine | TABLET, CHEWABLE;ORAL | 020154-006 | Oct 28, 1999 | 5,616,566*PED | ⤷ Try a Trial |
| Bristol Myers Squibb | SUSTIVA | efavirenz | CAPSULE;ORAL | 020972-002 | Sep 17, 1998 | 5,519,021*PED | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-003 | Jun 29, 2006 | 9,101,621 | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-005 | Dec 21, 2011 | 9,155,730 | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-002 | Dec 27, 2005 | 9,056,120 | ⤷ Try a Trial |
| Bristol Myers Squibb | REVLIMID | lenalidomide | CAPSULE;ORAL | 021880-004 | Jun 29, 2006 | 6,315,720 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for BRISTOL MYERS SQUIBB drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | 2.5 mg and 20 mg | ➤ Subscribe | 2016-07-12 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2010-03-19 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2009-04-09 |
| ➤ Subscribe | Nasal Spray | 4 mg/spray | ➤ Subscribe | 2016-07-15 |
| ➤ Subscribe | Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2016-12-28 |
| ➤ Subscribe | Tablets | 20 mg, 50 mg, 70 mg and 100 mg | ➤ Subscribe | 2010-06-28 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-17 |
| ➤ Subscribe | Delayed-release Capsules | 200 mg, 250 mg and 400 mg | ➤ Subscribe | 2004-06-01 |
| ➤ Subscribe | Capsules | 5 mg, 10 mg and 15 mg | ➤ Subscribe | 2010-08-30 |
| ➤ Subscribe | Capsules | 300 mg | ➤ Subscribe | 2009-07-20 |
| ➤ Subscribe | Capsules | 200 mg | ➤ Subscribe | 2010-02-16 |
| ➤ Subscribe | Capsules | 50 mg, 100 mg and 200 mg | ➤ Subscribe | 2016-11-03 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-06-14 |
| ➤ Subscribe | Tablets | 30 mg | ➤ Subscribe | 2005-06-01 |
| ➤ Subscribe | Tablets | 80 mg and 140 mg | ➤ Subscribe | 2011-06-16 |
| ➤ Subscribe | Capsules | 25 mg | ➤ Subscribe | 2010-07-12 |
International Patents for Bristol Myers Squibb Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| South Korea | 20050010812 | ⤷ Try a Trial |
| Japan | 2015044862 | ⤷ Try a Trial |
| Croatia | P20040280 | ⤷ Try a Trial |
| Costa Rica | 10967 | ⤷ Try a Trial |
| Montenegro | 01937 | ⤷ Try a Trial |
| Taiwan | I653228 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bristol Myers Squibb Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2487163 | SPC/GB17/010 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: COBICISTAT OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF AND ATAZANAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR ATAZANAVIR SULFATE; REGISTERED: UK EU/1/15/1025/001 20150715; UK EU/1/15/1025/002 20150715 |
| 1427415 | 122011100050 | Germany | ⤷ Try a Trial | PRODUCT NAME: APIXABAN ODER EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/11/691/001-005 20110518 |
| 2782584 | 122021000080 | Germany | ⤷ Try a Trial | PRODUCT NAME: ZUSAMMENSETZUNG, DIE SOWOHL ESTRADIOL (17SS-ESTRADIOL), GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES, HYDRATS ODER SOLVATS DAVON (EINSCHLIESSLICH IN FORM EINES HEMIHYDRATS), ALS AUCH PROGESTERON ENTHAELT; NAT. REGISTRATION NO/DATE: 2205034.00.00 20210924; FIRST REGISTRATION: BELGIEN BE582231 20210406 |
| 2782584 | 132021000000197 | Italy | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOLO (17SS-ESTRADIOLO) IN PARTICOLARE NELLA FORMA EMIIDRATA, E PROGESTERONE COMPRENDENTI LE VARIE FORME DI ESTRADIOLO (17SS-ESTRADIOLO), QUALI LE FORME IDRATE E SOLVATATE, INCLUDENDO LA FORMA EMIIDRATA, ED I SUOI SALI.(BIJUVA); AUTHORISATION NUMBER(S) AND DATE(S): BE582231, 20210406;048335018 -048335020, 20210517 |
| 1169038 | 132013902119320 | Italy | ⤷ Try a Trial | PRODUCT NAME: DASATINIB E I SUOI SALI FARMACEUTICAMENTE ACCETTABILI(SPRYCEL); AUTHORISATION NUMBER(S) AND DATE(S): DA EU/1/06/363/001 A EU/I/06/363/015, 20061120 |
| 0582455 | 40/2000 | Austria | ⤷ Try a Trial | PRODUCT NAME: EFAVIRENZ, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES; NAT. REGISTRATION NO/DATE: EU/1/99/110/001-004 1/99/111/001-004 19990528; FIRST REGISTRATION: LI 54908 01, 54908 02, 54908 03 19981120 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.