Bayer Hlthcare Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BAYER HLTHCARE, and when can generic versions of BAYER HLTHCARE drugs launch?
BAYER HLTHCARE has fifty-six approved drugs.
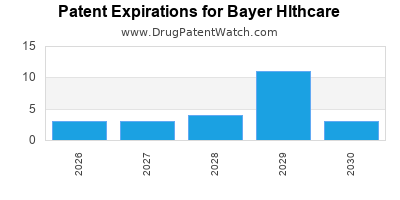
There are thirty-seven US patents protecting BAYER HLTHCARE drugs.
There are eight hundred and five patent family members on BAYER HLTHCARE drugs in fifty-eight countries and one hundred and forty-seven supplementary protection certificates in twenty countries.
Summary for Bayer Hlthcare
| International Patents: | 805 |
| US Patents: | 37 |
| Tradenames: | 57 |
| Ingredients: | 37 |
| NDAs: | 56 |
Drugs and US Patents for Bayer Hlthcare
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | VITRAKVI | larotrectinib sulfate | CAPSULE;ORAL | 210861-001 | Nov 26, 2018 | RX | Yes | No | 9,447,104 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bayer Hlthcare | CLIMARA | estradiol | FILM, EXTENDED RELEASE;TRANSDERMAL | 020375-002 | Dec 22, 1994 | AB2 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bayer Hlthcare | KERENDIA | finerenone | TABLET;ORAL | 215341-001 | Jul 9, 2021 | RX | Yes | No | 8,436,180 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Bayer Hlthcare | YASMIN | drospirenone; ethinyl estradiol | TABLET;ORAL-28 | 021098-001 | May 11, 2001 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bayer Hlthcare | ANGELIQ | drospirenone; estradiol | TABLET;ORAL | 021355-002 | Sep 28, 2005 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bayer Hlthcare | OSMOVIST 190 | iotrolan | INJECTABLE;INTRATHECAL | 019580-001 | Dec 7, 1989 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bayer Hlthcare | MYCELEX | clotrimazole | TROCHE/LOZENGE;ORAL | 018713-001 | Jun 17, 1983 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bayer Hlthcare
Paragraph IV (Patent) Challenges for BAYER HLTHCARE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 20 mg | ➤ Subscribe | 2009-03-05 |
| ➤ Subscribe | Tablets | 2.5 mg | ➤ Subscribe | 2009-09-04 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2014-02-28 |
| ➤ Subscribe | Tablets | 3 mg/0.03 mg/0.451 mg and 0.451 mg | ➤ Subscribe | 2012-09-28 |
| ➤ Subscribe | Transdermal System | 0.05 mg/day and 0.1 mg/day | ➤ Subscribe | 2005-09-12 |
| ➤ Subscribe | Tablets | 3 mg/0.02 mg | ➤ Subscribe | 2006-09-29 |
| ➤ Subscribe | Injection | 1.6 mg/mL | ➤ Subscribe | 2014-02-07 |
| ➤ Subscribe | Tablets | 0.5 mg, 1 mg,1.5 mg, 2 mg and2.5 mg | ➤ Subscribe | 2017-10-10 |
| ➤ Subscribe | Tablets | 0.25 mg/0.5 mg | ➤ Subscribe | 2015-01-08 |
| ➤ Subscribe | Tablets | 3 mg/0.02 mg/0.451 mg and 0.451 mg | ➤ Subscribe | 2012-11-13 |
| ➤ Subscribe | Tablets | 5 mg ad 10 mg | ➤ Subscribe | 2009-07-10 |
| ➤ Subscribe | Tablets | 3 mg; 2 mg/2 mg; 2 mg/3 mg; 1 mg | ➤ Subscribe | 2010-10-22 |
| ➤ Subscribe | Tablets | 25 mg, 50 mg and 100 mg | ➤ Subscribe | 2005-03-22 |
| ➤ Subscribe | Tablets | 40 mg | ➤ Subscribe | 2016-09-27 |
| ➤ Subscribe | Oral Suspension | 250 mg/5 mL and 500 mg/ 5 mL | ➤ Subscribe | 2009-10-16 |
| ➤ Subscribe | Tablets | 3 mg/0.03 mg | ➤ Subscribe | 2005-01-07 |
| ➤ Subscribe | Orally Disintegrating Tablets | 10 mg | ➤ Subscribe | 2011-12-22 |
| ➤ Subscribe | Tablets | 0.5 mg/1 mg | ➤ Subscribe | 2007-12-26 |
| ➤ Subscribe | Tablets | 3 mg/0.02 mg/0.451 mg and 0.451 mg | ➤ Subscribe | 2011-11-21 |
International Patents for Bayer Hlthcare Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Uruguay | 29861 | ⤷ Try a Trial |
| South Korea | 20180018827 | ⤷ Try a Trial |
| Guatemala | 200900057 | ⤷ Try a Trial |
| Peru | 20040197 | ⤷ Try a Trial |
| Taiwan | I341201 | ⤷ Try a Trial |
| Mexico | 356702 | ⤷ Try a Trial |
| Denmark | 2486942 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bayer Hlthcare Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1506193 | C01506193/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: RIOCIGUAT; REGISTRATION NO/DATE: SWISSMEDIC 62903 21.11.2013 |
| 0770388 | 9/2009 | Austria | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION AUS ESTRADIOLVALERAT UND DIENOGEST; NAT. REGISTRATION NO/DATE: 1-28003 20090203; FIRST REGISTRATION: BE BE 327792 20081103 |
| 2132206 | 2290018-7 | Sweden | ⤷ Try a Trial | PRODUCT NAME: FINERENONE AND ITS SALTS, SOLVATES AND SOLVATES OF THE SALT THEREOF; REG. NO/DATE: EU/1/21/1616 20220217 |
| 2782584 | LUC00245 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17SS-ESTRADIOL), EVENTUELLEMENT SOUS FORME D'UN SEL, HYDRATE OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI (Y COMPRIS SOUS FORME HEMIHYDRATEE), ET DE LA PROGESTERONE; AUTHORISATION NUMBER AND DATE: BE582231 20210701 |
| 1663978 | C01663978/01 | Switzerland | ⤷ Try a Trial | FORMER OWNER: BAYER HEALTHCARE LLC, US |
| 0770388 | 09C0018 | France | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL VALERATE; DIENOGEST; NAT. REGISTRATION NO/DATE: NL35170 20081210; FIRST REGISTRATION: BE327792 20081103 |
| 1663978 | SPC/GB13/076 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: REGORAFENIB AND ITS SALTS; REGISTERED: UK EU/1/13/858/001 20130829; UK EU/1/13/858/002 20130829 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.