Capecitabine - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for capecitabine and what is the scope of freedom to operate?
Capecitabine
is the generic ingredient in two branded drugs marketed by Accord Hlthcare, Alkem Labs Ltd, Amneal Pharms, Dr Reddys, Eugia Pharma, Hetero Labs Ltd V, Hikma, MSN, Reliance Life, Rising, Shilpa, Sun Pharm, Teva Pharms Usa, Teyro Labs, and Cheplapharm, and is included in fifteen NDAs. Additional information is available in the individual branded drug profile pages.There are twenty-eight drug master file entries for capecitabine. Twenty-four suppliers are listed for this compound.
Summary for capecitabine
| US Patents: | 0 |
| Tradenames: | 2 |
| Applicants: | 15 |
| NDAs: | 15 |
| Drug Master File Entries: | 28 |
| Finished Product Suppliers / Packagers: | 24 |
| Raw Ingredient (Bulk) Api Vendors: | 118 |
| Clinical Trials: | 1,741 |
| Patent Applications: | 2,715 |
| Formulation / Manufacturing: | see details |
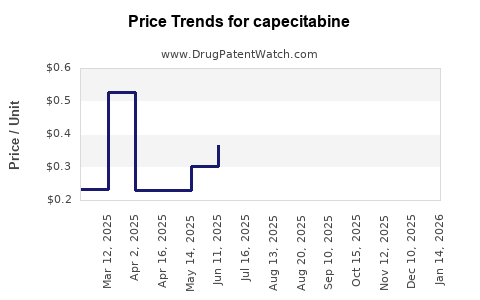
| Drug Prices: | Drug price trends for capecitabine |
| What excipients (inactive ingredients) are in capecitabine? | capecitabine excipients list |
| DailyMed Link: | capecitabine at DailyMed |
Recent Clinical Trials for capecitabine
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Chugai Pharmaceutical Co. | Phase 1 |
| University of Cincinnati | Phase 2/Phase 3 |
| CSPC ZhongQi Pharmaceutical Technology Co., Ltd. | Phase 3 |
Pharmacology for capecitabine
| Drug Class | Nucleoside Metabolic Inhibitor |
| Mechanism of Action | Nucleic Acid Synthesis Inhibitors |
Medical Subject Heading (MeSH) Categories for capecitabine
Paragraph IV (Patent) Challenges for CAPECITABINE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| XELODA | Tablets | capecitabine | 150 mg and 500 mg | 020896 | 1 | 2008-11-10 |
US Patents and Regulatory Information for capecitabine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dr Reddys | CAPECITABINE | capecitabine | TABLET;ORAL | 204345-002 | Dec 4, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Teva Pharms Usa | CAPECITABINE | capecitabine | TABLET;ORAL | 091649-001 | Sep 16, 2013 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Accord Hlthcare | CAPECITABINE | capecitabine | TABLET;ORAL | 202593-001 | Apr 23, 2015 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for capecitabine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Cheplapharm | XELODA | capecitabine | TABLET;ORAL | 020896-002 | Apr 30, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Cheplapharm | XELODA | capecitabine | TABLET;ORAL | 020896-002 | Apr 30, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Cheplapharm | XELODA | capecitabine | TABLET;ORAL | 020896-001 | Apr 30, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for capecitabine
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| medac Gesellschaft für klinische Spezialpräparate mbH | Capecitabine Medac | capecitabine | EMEA/H/C/002568 Capecitabine Medac is indicated for the adjuvant treatment of patients following surgery of stage-III (Dukes’ stage-C) colon cancer.Capecitabine Medac is indicated for the treatment of metastatic colorectal cancer.Capecitabine Medac is indicated for first-line treatment of advanced gastric cancer in combination with a platinum-based regimen.Capecitabine Medac in combination with docetaxel is indicated for the treatment of patients with locally advanced or metastatic breast cancer after failure of cytotoxic chemotherapy. Previous therapy should have included an anthracycline.Capecitabine Medac is also indicated as monotherapy for the treatment of patients with locally advanced or metastatic breast cancer after failure of taxanes and an anthracycline-containing chemotherapy regimen or for whom further anthracycline therapy is not indicated. |
Authorised | yes | no | no | 2012-11-19 | |
| Accord Healthcare S.L.U. | Capecitabine Accord | capecitabine | EMEA/H/C/002386 Capecitabine Accord is indicated for the adjuvant treatment of patients following surgery of stage-III (Dukes’ stage-C) colon cancer.Capecitabine Accord is indicated for the treatment of metastatic colorectal cancer.Capecitabine Accord is indicated for first-line treatment of advanced gastric cancer in combination with a platinum-based regimen.Capecitabine Accord in combination with docetaxel is indicated for the treatment of patients with locally advanced or metastatic breast cancer after failure of cytotoxic chemotherapy. Previous therapy should have included an anthracycline.Capecitabine Accord is also indicated as monotherapy for the treatment of patients with locally advanced or metastatic breast cancer after failure of taxanes and an anthracycline containing chemotherapy regimen or for whom further anthracycline therapy is not indicated. |
Authorised | yes | no | no | 2012-04-20 | |
| Teva Pharma B.V. | Capecitabine Teva | capecitabine | EMEA/H/C/002362 Capecitabine Teva is indicated for the adjuvant treatment of patients following surgery of stage III (Dukes’ stage C) colon cancer.Capecitabine Teva is indicated for the treatment of metastatic colorectal cancer.Capecitabine Teva is indicated for first‑line treatment of advanced gastric cancer in combination with a platinum‑based regimen.Capecitabine Teva in combination with docetaxel is indicated for the treatment of patients with locally advanced or metastatic breast cancer after failure of cytotoxic chemotherapy. Previous therapy should have included an anthracycline. Capecitabine Teva is also indicated as monotherapy for the treatment of patients with locally advanced or metastatic breast cancer after failure of taxanes and an anthracycline containing chemotherapy regimen or for whom further anthracycline therapy is not indicated. |
Authorised | yes | no | no | 2012-04-20 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.