Bupivacaine - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for bupivacaine and what is the scope of patent protection?
Bupivacaine
is the generic ingredient in sixteen branded drugs marketed by Pacira Pharms Inc, Innocoll, Innocoll Pharms, Civica, Eugia Pharma, Hikma Pharms, Hospira, Kindos, Somerset, Steriscience, Intl Medicated, Fresenius Kabi Usa, B Braun Medical Inc, Baxter Hlthcare Corp, Huons, Septodont, Amphastar Pharms Inc, and Heron Theraps Inc, and is included in forty-nine NDAs. There are thirty-four patents protecting this compound. Additional information is available in the individual branded drug profile pages.Bupivacaine has sixty-eight patent family members in thirty countries.
There are twelve drug master file entries for bupivacaine. Two suppliers are listed for this compound.
Summary for bupivacaine
| International Patents: | 68 |
| US Patents: | 34 |
| Tradenames: | 16 |
| Applicants: | 18 |
| NDAs: | 49 |
| Drug Master File Entries: | 12 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 125 |
| Clinical Trials: | 1,623 |
| Patent Applications: | 6,961 |
| Formulation / Manufacturing: | see details |
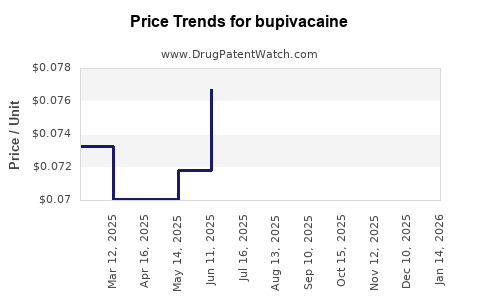
| Drug Prices: | Drug price trends for bupivacaine |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for bupivacaine |
| What excipients (inactive ingredients) are in bupivacaine? | bupivacaine excipients list |
| DailyMed Link: | bupivacaine at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for bupivacaine
Generic Entry Dates for bupivacaine*:
Constraining patent/regulatory exclusivity:
INDICATED TO PRODUCE POSTSURGICAL REGIONAL ANALGESIA IN ADULTS VIA ASCIATIC NERVE BLOCK IN THE POPLITEAL FOSSA AND VIA AN ADDUCTOR CANAL BLOCK Dosage:
INJECTABLE, LIPOSOMAL;INJECTION |
Generic Entry Dates for bupivacaine*:
Constraining patent/regulatory exclusivity:
Dosage:
SOLUTION, EXTENDED RELEASE;INFILTRATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for bupivacaine
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University Hospital "Sestre Milosrdnice" | N/A |
| National Trauma Center | N/A |
| Johnny K. Lee | Phase 4 |
Pharmacology for bupivacaine
| Drug Class | Amide Local Anesthetic |
| Physiological Effect | Local Anesthesia |
Medical Subject Heading (MeSH) Categories for bupivacaine
Paragraph IV (Patent) Challenges for BUPIVACAINE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| EXPAREL | Injectable Suspension | bupivacaine | 133 mg/10 mL | 022496 | 1 | 2021-12-28 |
| EXPAREL | Injectable Suspension | bupivacaine | 266 mg/20 mL | 022496 | 1 | 2021-08-20 |
US Patents and Regulatory Information for bupivacaine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heron Theraps Inc | ZYNRELEF KIT | bupivacaine; meloxicam | SOLUTION, EXTENDED RELEASE;PERIARTICULAR | 211988-001 | May 12, 2021 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Heron Theraps Inc | ZYNRELEF KIT | bupivacaine; meloxicam | SOLUTION, EXTENDED RELEASE;PERIARTICULAR | 211988-004 | May 12, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-001 | Oct 28, 2011 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Heron Theraps Inc | ZYNRELEF KIT | bupivacaine; meloxicam | SOLUTION, EXTENDED RELEASE;PERIARTICULAR | 211988-002 | May 12, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Steriscience | BUPIVACAINE HYDROCHLORIDE | bupivacaine hydrochloride | INJECTABLE;INJECTION | 091503-002 | Oct 18, 2011 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for bupivacaine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-002 | Oct 28, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-001 | Oct 28, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-002 | Oct 28, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-001 | Oct 28, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| Pacira Pharms Inc | EXPAREL | bupivacaine | INJECTABLE, LIPOSOMAL;INJECTION | 022496-002 | Oct 28, 2011 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for bupivacaine
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pacira Ireland Limited | Exparel liposomal | bupivacaine | EMEA/H/C/004586 Exparel liposomal is indicated:in adults as a brachial plexus block or femoral nerve block for treatment of post-operative pain.in adults and children aged 6 years or older as a field block for treatment of somatic post-operative pain from small- to medium-sized surgical wounds. |
Authorised | no | no | no | 2020-11-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for bupivacaine
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Spain | 2602273 | ⤷ Try a Trial | |
| Norway | 20071914 | ⤷ Try a Trial | |
| European Patent Office | 1809329 | COMPOSITION LOCALE ANESTHESIQUE A DELIVRANCE SOUTENUE CONTENANT AIBS (SUSTAINED LOCAL ANESTHETIC COMPOSITION CONTAINING SAIB) | ⤷ Try a Trial |
| Poland | 2767292 | ⤷ Try a Trial | |
| South Africa | 200702373 | Sustained local anesthetic composition containing preferably a sugar ester such as saib | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.