Albuterol sulfate - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for albuterol sulfate and what is the scope of patent protection?
Albuterol sulfate
is the generic ingredient in seventeen branded drugs marketed by Cipla, Lupin, Padagis Us, Sandoz, Teva Branded Pharm, Kindeva, Glaxosmithkline, Mylan Speciality Lp, Actavis Mid Atlantic, Apotex Inc, Bausch, Copley Pharm, Epic Pharma Llc, Landela Pharm, Luoxin Aurovitas, Mylan Speclt, Nephron, Ritedose Corp, Roxane, Sentiss, Sun Pharm, Teva Pharms, Watson Labs, Watson Labs Inc, Wockhardt Eu Operatn, Schering, Amneal Pharms, Chartwell Molecular, Chartwell Rx, Cosette, Hikma, Mova, Quagen, Teva, Rising, Muro, Strides Pharma, Aizant, Am Therap, Amneal Pharms Co, Aurobindo Pharma Ltd, Dash Pharms Natco, Eywa, Pliva, Sun Pharm Industries, Ucb Inc, Virtus Pharm, Warner Chilcott, Zydus Pharms, Astrazeneca, Boehringer Ingelheim, Fosun Pharma, and Watson Labs Teva, and is included in one hundred NDAs. There are thirty-five patents protecting this compound and one Paragraph IV challenge. Additional information is available in the individual branded drug profile pages.Albuterol sulfate has three hundred and sixty-three patent family members in twenty-nine countries.
There are eleven drug master file entries for albuterol sulfate. Forty-two suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for albuterol sulfate
| International Patents: | 363 |
| US Patents: | 35 |
| Tradenames: | 17 |
| Applicants: | 53 |
| NDAs: | 100 |
| Drug Master File Entries: | 11 |
| Finished Product Suppliers / Packagers: | 42 |
| Raw Ingredient (Bulk) Api Vendors: | 81 |
| Clinical Trials: | 44 |
| Patent Applications: | 6,475 |
| Formulation / Manufacturing: | see details |
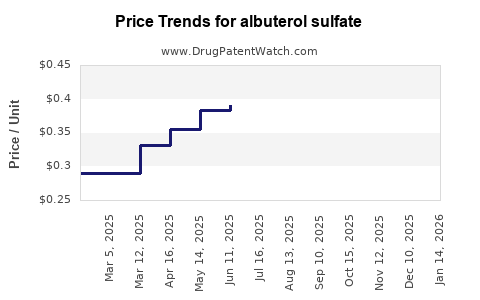
| Drug Prices: | Drug price trends for albuterol sulfate |
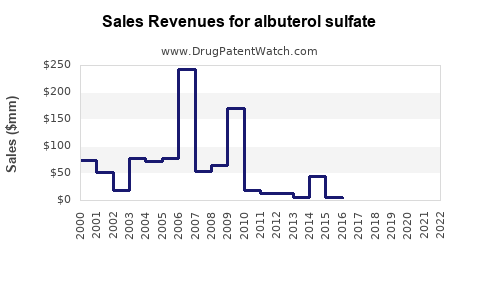
| Drug Sales Revenues: | Drug sales revenues for albuterol sulfate |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for albuterol sulfate |
| What excipients (inactive ingredients) are in albuterol sulfate? | albuterol sulfate excipients list |
| DailyMed Link: | albuterol sulfate at DailyMed |
Recent Clinical Trials for albuterol sulfate
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Parexel | Phase 3 |
| Bond Avillion 2 Development LP | Phase 3 |
| Aurobindo Pharma Ltd | Phase 3 |
Generic filers with tentative approvals for ALBUTEROL SULFATE
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Try a Trial | ⤷ Try a Trial | EQ 0.083% BASE;0.017% | SOLUTION;INHALATION |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Pharmacology for albuterol sulfate
| Drug Class | beta2-Adrenergic Agonist |
| Mechanism of Action | Adrenergic beta2-Agonists |
Paragraph IV (Patent) Challenges for ALBUTEROL SULFATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PROVENTIL-HFA | Inhalation Aerosol | albuterol sulfate | 0.09 mg base per actuation | 020503 | 1 | 2015-05-20 |
| PROAIR HFA | Inhalation Aerosol | albuterol sulfate | 0.09 mg base per actuation | 021457 | 1 | 2012-05-18 |
| ACCUNEB | Inhalation Solution | albuterol sulfate | 0.021% | 020949 | 1 | 2005-10-19 |
| ACCUNEB | Inhalation Solution | albuterol sulfate | 0.042% | 020949 | 1 | 2004-04-06 |
US Patents and Regulatory Information for albuterol sulfate
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Eywa | ALBUTEROL SULFATE | albuterol sulfate | TABLET;ORAL | 213524-002 | Oct 8, 2020 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva Branded Pharm | PROAIR HFA | albuterol sulfate | AEROSOL, METERED;INHALATION | 021457-001 | Oct 29, 2004 | AB2 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Ucb Inc | ALBUTEROL SULFATE | albuterol sulfate | TABLET;ORAL | 073121-001 | Sep 29, 1992 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Epic Pharma Llc | ALBUTEROL SULFATE | albuterol sulfate | SOLUTION;INHALATION | 075063-001 | Feb 9, 1999 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for albuterol sulfate
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Teva Branded Pharm | PROAIR DIGIHALER | albuterol sulfate | POWDER, METERED;INHALATION | 205636-002 | Dec 21, 2018 | ⤷ Try a Trial | ⤷ Try a Trial |
| Teva Branded Pharm | PROAIR HFA | albuterol sulfate | AEROSOL, METERED;INHALATION | 021457-001 | Oct 29, 2004 | ⤷ Try a Trial | ⤷ Try a Trial |
| Teva Branded Pharm | PROAIR HFA | albuterol sulfate | AEROSOL, METERED;INHALATION | 021457-001 | Oct 29, 2004 | ⤷ Try a Trial | ⤷ Try a Trial |
| Schering | PROVENTIL | albuterol sulfate | SYRUP;ORAL | 018062-001 | Jan 19, 1983 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for albuterol sulfate
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hong Kong | 1223311 | 用於吸入器的劑量計數器、吸入器及其軸 (DOSE COUNTERS FOR INHALERS, INHALERS AND SHAFTS THEREOF) | ⤷ Try a Trial |
| Brazil | 112017003865 | inalador com módulo de verificação de conformidade com um limiar predeterminado. | ⤷ Try a Trial |
| European Patent Office | 3097937 | MODULE DE SURVEILLANCE DE LA CONFORMITÉ POUR UN INHALATEUR ACTIONNÉ PAR LA RESPIRATION (COMPLIANCE MONITORING MODULE FOR A BREATH-ACTUATED INHALER) | ⤷ Try a Trial |
| South Korea | 101618933 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.