Ucb Inc Company Profile
✉ Email this page to a colleague
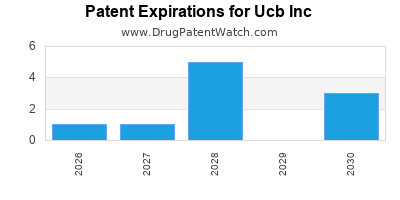
What is the competitive landscape for UCB INC, and when can generic versions of UCB INC drugs launch?
UCB INC has forty-two approved drugs.
There are thirty-four US patents protecting UCB INC drugs.
There are four hundred and eighty-four patent family members on UCB INC drugs in fifty-one countries and thirty-nine supplementary protection certificates in fourteen countries.
Summary for Ucb Inc
| International Patents: | 484 |
| US Patents: | 34 |
| Tradenames: | 29 |
| Ingredients: | 24 |
| NDAs: | 42 |
| Drug Master File Entries: | 1 |
| Patent Litigation for Ucb Inc: | See patent lawsuits for Ucb Inc |
Drugs and US Patents for Ucb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-001 | May 12, 2016 | RX | Yes | No | 6,911,461 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Ucb Inc | FLUXID | famotidine | TABLET, ORALLY DISINTEGRATING;ORAL | 021712-001 | Sep 24, 2004 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-002 | May 12, 2016 | RX | Yes | No | 10,729,653 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-003 | May 12, 2016 | RX | Yes | No | 6,911,461 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Ucb Inc | AMITRIPTYLINE HYDROCHLORIDE | amitriptyline hydrochloride | TABLET;ORAL | 085935-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Ucb Inc | NEUPRO | rotigotine | FILM, EXTENDED RELEASE;TRANSDERMAL | 021829-002 | May 9, 2007 | RX | Yes | No | 9,925,150 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Ucb Inc | KEPPRA | levetiracetam | TABLET;ORAL | 021035-001 | Nov 30, 1999 | AB | RX | Yes | No | 8,802,142*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Ucb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ucb Inc | KEPPRA | levetiracetam | TABLET;ORAL | 021035-003 | Nov 30, 1999 | 4,837,223 | ⤷ Try a Trial |
| Ucb Inc | UNIVASC | moexipril hydrochloride | TABLET;ORAL | 020312-001 | Apr 19, 1995 | 4,743,450 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | SOLUTION;INTRAVENOUS | 022254-001 | Oct 28, 2008 | 5,654,301 | ⤷ Try a Trial |
| Ucb Inc | NEUPRO | rotigotine | FILM, EXTENDED RELEASE;TRANSDERMAL | 021829-004 | Apr 2, 2012 | 6,884,434 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | SOLUTION;ORAL | 022255-001 | Apr 20, 2010 | 5,654,301 | ⤷ Try a Trial |
| Ucb Inc | VIMPAT | lacosamide | TABLET;ORAL | 022253-004 | Oct 28, 2008 | 5,654,301 | ⤷ Try a Trial |
| Ucb Inc | UNIVASC | moexipril hydrochloride | TABLET;ORAL | 020312-002 | Apr 19, 1995 | 4,743,450 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for UCB INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 50 mg, 100 mg, 150 mg, and 200 mg | ➤ Subscribe | 2012-10-29 |
| ➤ Subscribe | Extended-release Tablets | 1000 mg | ➤ Subscribe | 2011-01-07 |
| ➤ Subscribe | Tablets | 7.5mg/12.5mg 15 mg/25 mg and 15 mg/12.5 mg | ➤ Subscribe | 2004-01-15 |
| ➤ Subscribe | Extended-release Transdermal Film | 1 mg/24 hr, 2 mg/24 hr, 3 mg/24 hr,4 mg/24 hr,6 mg/24 hr, and 8 mg/24 hr | ➤ Subscribe | 2013-11-26 |
| ➤ Subscribe | Oral Solution | 10 mg/mL | ➤ Subscribe | 2012-10-29 |
| ➤ Subscribe | Injection | 10 mg/mL, 20 mL | ➤ Subscribe | 2016-06-30 |
| ➤ Subscribe | Orally Disintegrating Tablets | 0.25 mg, 0.5 mg, 1 mg and 2 mg | ➤ Subscribe | 2005-12-27 |
| ➤ Subscribe | Tablets | 1000 mg | ➤ Subscribe | 2007-01-24 |
International Patents for Ucb Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 2021000482 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2011076879 | ⤷ Try a Trial |
| China | 1671365 | ⤷ Try a Trial |
| Spain | 2679800 | ⤷ Try a Trial |
| Norway | 324051 | ⤷ Try a Trial |
| Poland | 2515887 | ⤷ Try a Trial |
| Russian Federation | 2733720 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Ucb Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1452524 | 10/2016 | Austria | ⤷ Try a Trial | PRODUCT NAME: BRIVARACETAM; REGISTRATION NO/DATE: EU/1/15/1073 (MITTEILUNG) 20160118 |
| 1429780 | 13C0012 | France | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE CIPROFLOXACINE ET DE DEXAMETHASONE, EN PARTICULIER DE CHLORHYDRATE DE CIPROFLOXACINE ET DE DEXAMETHASONE; NAT. REGISTRATION NO/DATE: NL 41308 20121214; FIRST REGISTRATION: 48976 20120808 |
| 1033978 | 06C0025 | France | ⤷ Try a Trial | PRODUCT NAME: ROTIGOTINE; REGISTRATION NO/DATE: EU/1/05/331/001 20060215 |
| 1452524 | 132016000045699 | Italy | ⤷ Try a Trial | PRODUCT NAME: BRIVARACETAM(BRIVIACT); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/15/1073, 20160118 |
| 1452524 | 530 | Finland | ⤷ Try a Trial | |
| 0888289 | C300376 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LACOSAMIDE; REGISTRATION NO/DATE: EU/1/08/470/001-016 20080829 |
| 1110543 | SPC/GB08/005 | United Kingdom | ⤷ Try a Trial | SUPPLEMENTARY PROTECTION CERTIFICATE NO SPC/GB08/005 GRANTED TO MERCK SHARP + DOHME CORP. IN RESPECT OF THE PRODUCT DESLORATADINE OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, IN COMBINATION WITH PSEUDOEPHEDRINE OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR ESTER, THE GRANT OF WHICH WAS ADVERTISED IN JOURNAL NO 6322 DATED 21 JULY 2010 HAS HAD ITS MAXIMUM PERIOD OF DURATION CORRECTED, SUBJECT TO THE PAYMENT OF THE PRESCRIBED FEES IT WILL EXPIRE ON 31 JULY 2022. |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.