Par Pharm Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PAR PHARM INC, and what generic alternatives to PAR PHARM INC drugs are available?
PAR PHARM INC has thirty-eight approved drugs.
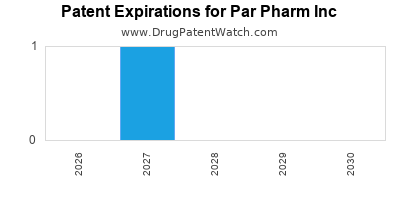
There is one US patent protecting PAR PHARM INC drugs. There are seven tentative approvals on PAR PHARM INC drugs.
There are nine patent family members on PAR PHARM INC drugs in eight countries and sixty-one supplementary protection certificates in fourteen countries.
Drugs and US Patents for Par Pharm Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Par Pharm Inc | DEXMETHYLPHENIDATE HYDROCHLORIDE | dexmethylphenidate hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 202842-008 | Nov 30, 2016 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | PRAZIQUANTEL | praziquantel | TABLET;ORAL | 208820-001 | Nov 27, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Par Pharm Inc | ALOSETRON HYDROCHLORIDE | alosetron hydrochloride | TABLET;ORAL | 206113-002 | Feb 23, 2018 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Par Pharm Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 1948613 | ⤷ Try a Trial |
| Australia | 2006240490 | ⤷ Try a Trial |
| Mexico | 2007013118 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Par Pharm Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0503785 | C300375 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN OLMESARTAN MEDOXOMIL, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT EN AMLODIPINEBESILAAT; REGISTRATION NO/DATE: RVG100984, RVG100986-87, RVG100989-91, RVG100993-95 20080819 |
| 2330892 | SPC/GB17/001 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: GLYCEROL PHENYLBUTYRATE; REGISTERED: UK EU/1/15/1062 20151201; UK PLGB53487/0001 20151201 |
| 1915993 | 300625 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE BEVATTENDE ALISKIREN, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, EN AMLODIPINE, OF EEN FARMACEUATISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/11/686/001-056 20110414 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.