Emd Serono Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for EMD SERONO INC, and what generic alternatives to EMD SERONO INC drugs are available?
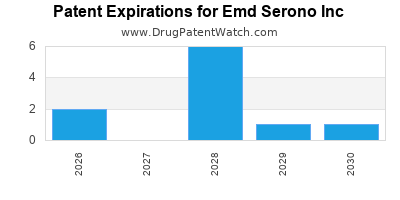
EMD SERONO INC has six approved drugs.
There are thirteen US patents protecting EMD SERONO INC drugs.
There are one hundred and ninety-five patent family members on EMD SERONO INC drugs in forty-two countries and eighty-nine supplementary protection certificates in sixteen countries.
Summary for Emd Serono Inc
| International Patents: | 195 |
| US Patents: | 13 |
| Tradenames: | 6 |
| Ingredients: | 5 |
| NDAs: | 6 |
| Drug Master File Entries: | 2 |
Drugs and US Patents for Emd Serono Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Emd Serono Inc | TEPMETKO | tepotinib hydrochloride | TABLET;ORAL | 214096-001 | Feb 3, 2021 | RX | Yes | Yes | 8,580,781 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Emd Serono Inc | GLUCOPHAGE | metformin hydrochloride | TABLET;ORAL | 020357-004 | Nov 5, 1998 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Emd Serono Inc | MAVENCLAD | cladribine | TABLET;ORAL | 022561-001 | Mar 29, 2019 | RX | Yes | Yes | 10,849,919 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Emd Serono Inc | GLUCOPHAGE | metformin hydrochloride | TABLET;ORAL | 020357-001 | Mar 3, 1995 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Emd Serono Inc | MAVENCLAD | cladribine | TABLET;ORAL | 022561-001 | Mar 29, 2019 | RX | Yes | Yes | 7,713,947 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Emd Serono Inc | GLUCOPHAGE | metformin hydrochloride | TABLET;ORAL | 020357-003 | Nov 5, 1998 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Emd Serono Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Emd Serono Inc | CETROTIDE | cetrorelix acetate | POWDER;SUBCUTANEOUS | 021197-001 | Aug 11, 2000 | 7,605,121 | ⤷ Try a Trial |
| Emd Serono Inc | CETROTIDE | cetrorelix acetate | POWDER;SUBCUTANEOUS | 021197-001 | Aug 11, 2000 | 6,319,192 | ⤷ Try a Trial |
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-002 | Sep 26, 1997 | 4,517,181 | ⤷ Try a Trial |
| Emd Serono Inc | GLUCOPHAGE XR | metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 021202-001 | Oct 13, 2000 | 6,475,521 | ⤷ Try a Trial |
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-002 | Sep 26, 1997 | 4,703,035 | ⤷ Try a Trial |
| Emd Serono Inc | GEREF | sermorelin acetate | INJECTABLE;INJECTION | 020443-001 | Sep 26, 1997 | 4,517,181 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Emd Serono Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Serbia | 20050735 | ⤷ Try a Trial |
| Canada | 2520522 | ⤷ Try a Trial |
| Spain | 2349136 | ⤷ Try a Trial |
| Eurasian Patent Organization | 009714 | ⤷ Try a Trial |
| Malaysia | 163477 | ⤷ Try a Trial |
| Cyprus | 1112614 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Emd Serono Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1506211 | CR 2014 00037 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ET KOMBINATIONSPRODUKT AF DAPAGLIFLOZIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF, HERUNDER DAPAGLIFLOZINPROPANDIOLMONOHYDRAT OG METFORMIN ELLER SALTE DERAF, HERUNDER METFORMINHYDROCHLORID; REG. NO/DATE: EU/1/13/900 20140116 |
| 1506211 | 92496 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE DAPAGLIFLOZINE OU D UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI ET DE METFORMINE OU D UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI,TELLE QUE PROTEGEE PAR LE BREVET DE BASE EP1506211 B1 |
| 1412357 | C300357 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: SITAGLIPTINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVA ARDBAAR ZOUT, IN HET BIJZONDER HET MONOFOSFAAT, EN METFORMINE DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER HET HYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/08/455/001-014 20080716 |
| 1506211 | 42/2014 | Austria | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION VON DAPAGLIFLOZIN ODER EINES PHARMAZEUTISCH VERTRAEGLICHEN SALZES DAVON UND METFORMIN UND EINES PHARMAZEUTISCH VERTRAEGLICHEN SALZES DAVON; REGISTRATION NO/DATE: EU/1/13/900 (MITTEILUNG) 20140121 |
| 2498758 | 2090013-0 | Sweden | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF METFORMIN HYDROCHLORIDE, SAXAGLIPTIN OR A PHARMACEUTICALLY ACCEPABLE SALT THEREOF, AND DAPAGLIFLOZIN OR PH ARMACEUTICALLY ACCEPTABLE SOLVATE THEREOF; REG. NO/DATE: EU/1/19/1401 20191113 |
| 1730131 | C01730131/02 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: EMPAGLIFLOZIN UND METFORMINHYDROCHLORID; REGISTRATION NO/DATE: SWISSMEDIC 65570 12.11.2015 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.