Bausch And Lomb Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BAUSCH AND LOMB INC, and when can generic versions of BAUSCH AND LOMB INC drugs launch?
BAUSCH AND LOMB INC has nineteen approved drugs.
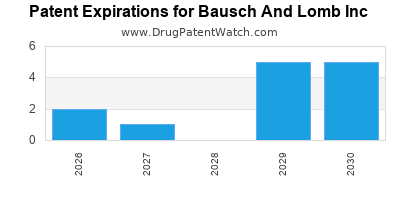
There are thirty-two US patents protecting BAUSCH AND LOMB INC drugs.
There are three hundred and seventy-four patent family members on BAUSCH AND LOMB INC drugs in thirty-one countries and forty-one supplementary protection certificates in fourteen countries.
Summary for Bausch And Lomb Inc
| International Patents: | 374 |
| US Patents: | 32 |
| Tradenames: | 19 |
| Ingredients: | 15 |
| NDAs: | 19 |
| Drug Master File Entries: | 1 |
Drugs and US Patents for Bausch And Lomb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bausch And Lomb Inc | ALAWAY | ketotifen fumarate | SOLUTION/DROPS;OPHTHALMIC | 208158-001 | Sep 24, 2020 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bausch And Lomb Inc | XIIDRA | lifitegrast | SOLUTION/DROPS;OPHTHALMIC | 208073-001 | Jul 11, 2016 | AB | RX | Yes | Yes | 9,085,553 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Bausch And Lomb Inc | XIIDRA | lifitegrast | SOLUTION/DROPS;OPHTHALMIC | 208073-001 | Jul 11, 2016 | AB | RX | Yes | Yes | 7,314,938 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | |
| Bausch And Lomb Inc | XIPERE | triamcinolone acetonide | SUSPENSION;INJECTION | 211950-001 | Oct 22, 2021 | RX | Yes | Yes | 8,636,713 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch And Lomb Inc | TIMOPTIC | timolol maleate | SOLUTION/DROPS;OPHTHALMIC | 018086-001 | Approved Prior to Jan 1, 1982 | AT1 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch And Lomb Inc | XIIDRA | lifitegrast | SOLUTION/DROPS;OPHTHALMIC | 208073-001 | Jul 11, 2016 | AB | RX | Yes | Yes | 9,447,077 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Bausch And Lomb Inc | LOTEMAX | loteprednol etabonate | GEL;OPHTHALMIC | 202872-001 | Sep 28, 2012 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bausch And Lomb Inc
Paragraph IV (Patent) Challenges for BAUSCH AND LOMB INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Ophthalmic Solution | 1.5% | ➤ Subscribe | 2013-09-09 |
International Patents for Bausch And Lomb Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Eurasian Patent Organization | 201592109 | ⤷ Try a Trial |
| Canada | 2975106 | ⤷ Try a Trial |
| Hungary | E041945 | ⤷ Try a Trial |
| Germany | 202013012742 | ⤷ Try a Trial |
| Japan | 6900442 | ⤷ Try a Trial |
| Japan | 2018162313 | ⤷ Try a Trial |
| South Korea | 20150100763 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bausch And Lomb Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1586316 | C300494 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BROMFENAC, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF HYDRAAT; REGISTRATION NO/DATE: EU/1/11/692/001 20110518 |
| 3043773 | 21C1057 | France | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE OU L'UN DE SES SELS AVEC OLOPATADINE OU L'UN DE SES SELS; NAT. REGISTRATION NO/DATE: NL52121 20211026; FIRST REGISTRATION: AT - 140638 20210426 |
| 2826776 | CR 2021 00013 | Denmark | ⤷ Try a Trial | PRODUCT NAME: EN KOMBINATION AF (A) (R)-1-(2,2-DIFLUOROBENZO(D)(1,3)DIOXOL-5-YL)-N-(1-(2,3-DIHYDROXYPROPYL)-6-FLUORO-2-(1-HYDROXY-2-METHYLPROPAN-2-YL)-1H-INDOL-5-YL)CYCLOPROPANECARBOXAMID ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF OG (B) ....; REG. NO/DATE: EU/1/18/1306 20181106 |
| 1586316 | 122011100019 | Germany | ⤷ Try a Trial | PRODUCT NAME: BROMFENAC (2-AMINO-3-(4-BROMOBENZOYL)PHENYLESSIGSAEURE); REGISTRATION NO/DATE: EU/1/11/692/001 20110518 |
| 2826776 | 2021C/517 | Belgium | ⤷ Try a Trial | PRODUCT NAME: SYMKEVI - TEZACAFTOR/IVACAFTOR; EEN COMBINATIE VAN (A) (R)-1-(2,2-DIFLUOROBENZO(D)(1,3)DIOXOL-5-YL)-N-(1-(2,3-DIHYDROXYPROPYL)-6-FLUORO-2-(1-HYDROXY-2-METHYLPROPAN-2-YL)-1H-INDOL-5-YL)CYCLOPROPANECARBOXAMIDE OF EEN VANUIT FARMACEUTISCH OOGPUNT GESCHIKT ZOUT DAARVAN EN (B) N-(5-HYDROXY-2,4-DITERT-BUTYL-PHENYL)-4-OXO-1H-QUINOLINE-3-CARBOXAMIDE OF EEN VANUIT FARMACEUTISCH OOGPUNT GESCHIKT ZOUT DAARVAN; AUTHORISATION NUMBER AND DATE: EU/1/18/1306 20181106 |
| 0957929 | PA2006004 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: PEGAPTANIBUM; REG. NO/DATE: EU/1/05/325/001 20060131 |
| 0957929 | SPC/GB06/021 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: PEGAPTANIB, PREFERABLY IN THE FORM OF ITS SODIUM SALT; REGISTERED: UK EU/1/05/325/001 20060201 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.