ZYDELIG Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Zydelig, and what generic alternatives are available?
Zydelig is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and eleven patent family members in forty countries.
The generic ingredient in ZYDELIG is idelalisib. There are two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the idelalisib profile page.
DrugPatentWatch® Generic Entry Outlook for Zydelig
Zydelig was eligible for patent challenges on July 23, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be September 2, 2033. This may change due to patent challenges or generic licensing.
There is one Paragraph IV patent challenge for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
Summary for ZYDELIG
| International Patents: | 111 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 106 |
| Clinical Trials: | 13 |
| Patent Applications: | 1,009 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for ZYDELIG |
| What excipients (inactive ingredients) are in ZYDELIG? | ZYDELIG excipients list |
| DailyMed Link: | ZYDELIG at DailyMed |
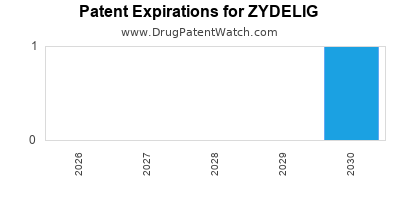

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ZYDELIG
Generic Entry Date for ZYDELIG*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ZYDELIG
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Loxo Oncology, Inc. | Phase 3 |
| Prospect Creek Foundation | Phase 1 |
| Oregon Health and Science University | Phase 1 |
Pharmacology for ZYDELIG
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 3A Inhibitors Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ZYDELIG
Paragraph IV (Patent) Challenges for ZYDELIG
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ZYDELIG | Tablets | idelalisib | 100 mg and 150 mg | 205858 | 1 | 2022-03-23 |
US Patents and Regulatory Information for ZYDELIG
ZYDELIG is protected by nine US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ZYDELIG is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting ZYDELIG
Polymorphic forms of (S)-2-(1-(9H-purin-6-ylamino)propyl)-5-fluoro-3-phenylquinazolin-4(3H)-on- e
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Polymorphic forms of (S)-2-(1-(9H-purin-6-ylamino)propyl)-5-fluoro-3-phenylquinazolin-4(3H)-on- e
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: FOR THE TREATMENT OF PATIENTS WITH CLL, FL, OR SLL
5-fluoro-3-phenyl-2[1-(9H-purin-6-ylamino)propyl]-3H-quinazolin-4-one and 6-fluoro-3-phenyl-2-[1-(9H-purin-6-ylamino)ethyl]-3H-quinazolin-4-one as inhibitors of human phosphatidylinositol 3-kinase delta
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: FOR THE TREATMENT OF PATIENTS WITH CLL, FL, OR SLL
5-fluoro-3-phenyl-2-[1-(9h-purin-6-ylamino)propyl]-3h-quinazolin-4-one as an inhibitor of human phosphatidylinositol 3-kinase delta
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: INHIBITION ON PI3K KINASE
Polymorphic forms of (S)-2-(1-(9H-purin-6-ylamino)propyl)-5-fluoro-3-phenylquinazolin-4(3H)-on- e
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Therapies for hematologic malignancies
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: IN COMBINATION WITH RITUXIMAB, FOR THE TREATMENT OF PATIENTS WITH RELAPSED CHRONIC LYMPHOCYTIC LEUKEMIA (CLL)
Quinazolinones as inhibitors of human phosphatidylinositol 3-kinase delta
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: FOR THE TREATMENT OF PATIENTS WITH RELAPSED FOLLICULAR B-CELL NON-HODGKIN LYMPHOMA OR [RELAPSED] SMALL LYMPHOCYTIC LYMPHOMA
Quinazolinones as inhibitors of human phosphatidylinositol 3-kinase delta
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: FOR THE TREATMENT OF PATIENTS WITH CLL, FL, OR SLL
Quinazolinones as inhibitors of human phosphatidylinositol 3-kinase delta
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ZYDELIG
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-001 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | ZYDELIG | idelalisib | TABLET;ORAL | 205858-002 | Jul 23, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for ZYDELIG
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Ireland UC | Zydelig | idelalisib | EMEA/H/C/003843 Zydelig is indicated in combination with an anti‑CD20 monoclonal antibody (rituximab or ofatumumab) for the treatment of adult patients with chronic lymphocytic leukaemia (CLL):who have received at least one prior therapy, oras first line treatment in the presence of 17p deletion or TP53 mutation in patients who are not eligible for any other therapies.Zydelig is indicated as monotherapy for the treatment of adult patients with follicular lymphoma (FL) that is refractory to two prior lines of treatment. |
Authorised | no | no | no | 2014-09-18 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ZYDELIG
When does loss-of-exclusivity occur for ZYDELIG?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 0253
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 13203620
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2014021935
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 64305
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 14002358
Estimated Expiration: ⤷ Try a Trial
China
Patent: 4334560
Estimated Expiration: ⤷ Try a Trial
Patent: 6146506
Estimated Expiration: ⤷ Try a Trial
Colombia
Patent: 71131
Estimated Expiration: ⤷ Try a Trial
Costa Rica
Patent: 140460
Estimated Expiration: ⤷ Try a Trial
Ecuador
Patent: 14020478
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 5407
Estimated Expiration: ⤷ Try a Trial
Patent: 1491473
Estimated Expiration: ⤷ Try a Trial
Patent: 1690461
Estimated Expiration: ⤷ Try a Trial
Patent: 1691327
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 34241
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 06345
Estimated Expiration: ⤷ Try a Trial
India
Patent: 05DEN2014
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 15509537
Estimated Expiration: ⤷ Try a Trial
Patent: 16104823
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 14010656
Estimated Expiration: ⤷ Try a Trial
Moldova, Republic of
Patent: 140100
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 379
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 9684
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 141792
Estimated Expiration: ⤷ Try a Trial
Philippines
Patent: 014501920
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 34241
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 34241
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 201405446P
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 34241
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1405870
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 140133590
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 48273
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 1350486
Estimated Expiration: ⤷ Try a Trial
Uruguay
Patent: 656
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ZYDELIG around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 03035075 | ⤷ Try a Trial | |
| Japan | 2015155436 | 血液学的な悪性疾患のための療法 (THERAPY FOR HEMATOLOGIC MALIGNANCY) | ⤷ Try a Trial |
| South Korea | 20110105771 | THERAPIES FOR HEMATOLOGIC MALIGNANCIES | ⤷ Try a Trial |
| Netherlands | 300867 | ⤷ Try a Trial | |
| Philippines | 12014501920 | POLYMORPHIC FORMS OF (S)-2-(1-(9H-PURIN-6-YLAMINO)PROPYL)-5-FLUORO-3-PHENYLQUINAZOLIN-4(3H)-ONE | ⤷ Try a Trial |
| Luxembourg | C00005 | ⤷ Try a Trial | |
| China | 104042618 | Therapies For Hematologic Malignancies | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ZYDELIG
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1761540 | 2017/006 | Ireland | ⤷ Try a Trial | PRODUCT NAME: IDELALISIB OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; REGISTRATION NO/DATE: EU/1/14/938 20140918 |
| 1761540 | 9/2017 | Austria | ⤷ Try a Trial | PRODUCT NAME: IDELALISIB ODER EIN PHARMAZEUTISCH UNBEDENKLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/14/938 (MITTEILUNG) 20140919 |
| 1761540 | LUC00005 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: IDELALISIB OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/14/938 20140919 |
| 1761540 | PA2017004,C1761540 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: IDELALISIBAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; REGISTRATION NO/DATE: EU/1/14/938 20140918 |
| 1761540 | 17C1007 | France | ⤷ Try a Trial | PRODUCT NAME: IDELALISIB OU SEL DE CELUI-CI ACCEPTABLE D'UN POINT DE VUE PHARMACEUTIQUE; REGISTRATION NO/DATE: EU/1/14/938 20140919 |
| 1761540 | CR 2017 00007 | Denmark | ⤷ Try a Trial | PRODUCT NAME: IDELALISIB ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/14/938 20140919 |
| 1761540 | PA2017004 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: IDELALISIBAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; REGISTRATION NO/DATE: EU/1/14/938 20140918 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


