VIREAD Drug Patent Profile
✉ Email this page to a colleague
When do Viread patents expire, and when can generic versions of Viread launch?
Viread is a drug marketed by Gilead Sciences Inc and is included in two NDAs.
The generic ingredient in VIREAD is tenofovir disoproxil fumarate. There are forty-two drug master file entries for this compound. Fifteen suppliers are listed for this compound. Additional details are available on the tenofovir disoproxil fumarate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Viread
A generic version of VIREAD was approved as tenofovir disoproxil fumarate by TEVA PHARMS USA on March 18th, 2015.
Summary for VIREAD
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 89 |
| Clinical Trials: | 91 |
| Patent Applications: | 4,317 |
| Formulation / Manufacturing: | see details |
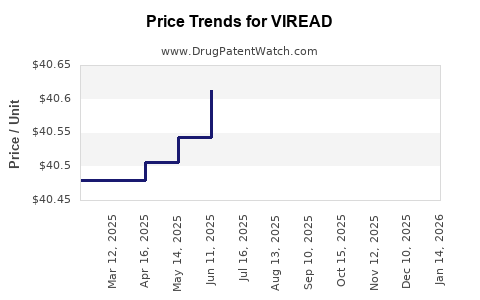
| Drug Prices: | Drug price information for VIREAD |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VIREAD |
| What excipients (inactive ingredients) are in VIREAD? | VIREAD excipients list |
| DailyMed Link: | VIREAD at DailyMed |
Recent Clinical Trials for VIREAD
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Doris Duke Charitable Foundation | Phase 4 |
| Albert Einstein College of Medicine | Phase 4 |
| Université Protestant au Congo | Phase 4 |
Pharmacology for VIREAD
Anatomical Therapeutic Chemical (ATC) Classes for VIREAD
Paragraph IV (Patent) Challenges for VIREAD
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| VIREAD | Tablets | tenofovir disoproxil fumarate | 150 mg, 200 mg, and 250 mg | 021356 | 1 | 2012-05-17 |
| VIREAD | Tablets | tenofovir disoproxil fumarate | 300 mg | 021356 | 1 | 2010-01-26 |
US Patents and Regulatory Information for VIREAD
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | POWDER;ORAL | 022577-001 | Jan 18, 2012 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-004 | Jan 18, 2012 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-002 | Jan 18, 2012 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-003 | Jan 18, 2012 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-001 | Oct 26, 2001 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for VIREAD
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-003 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-002 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | POWDER;ORAL | 022577-001 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | TABLET;ORAL | 021356-004 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | POWDER;ORAL | 022577-001 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| Gilead Sciences Inc | VIREAD | tenofovir disoproxil fumarate | POWDER;ORAL | 022577-001 | Jan 18, 2012 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for VIREAD
See the table below for patents covering VIREAD around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 1251679 | ⤷ Try a Trial | |
| Japan | 2015199773 | ヌクレオチドアナログ組成物および合成方法 (NUCLEOTIDE ANALOG COMPOSITION AND SYNTHESIS METHOD) | ⤷ Try a Trial |
| Netherlands | 300344 | ⤷ Try a Trial | |
| Japan | 3561272 | ⤷ Try a Trial | |
| South Korea | 100619298 | ⤷ Try a Trial | |
| Germany | 69324923 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VIREAD
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1663240 | 122015000087 | Germany | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION VON RILPIVIRIN ODER EINER THERAPEUTISCH AEQUIVALENTEN FORM DAVON, DIE DURCH DAS GRUNDPATENT GESCHUETZT IST, WIE Z.B EIN PHARMAZEUTISCH VERTRAEGLICHES SALZ VON RILPIVIRIN, EINSCHLIESSLICH DES HYDROCHLORIDSALZES VON RILPIVIRIN, UND TENOFOVIR, INSBESONDERE TENOFOVIRDISOPROXILFUMARAT; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 1663240 | 375 5032-2016 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: RILPIVIRIN VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM/EMTRICITABIN/TENOFOVIR-ALAFENAMID VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/16/1112 20160623 |
| 2487166 | 2017003 | Norway | ⤷ Try a Trial | PRODUCT NAME: KOBICISTAT ELLER FARMASOEYTISK AKSEPTABELT SALT DERAV OG TENOFOVIR ALAFENAMID ELLER FARMASOEYTISK AKSEPTABELT SALT DERAV, SPESIELT TENOFOVIR ALAFENAMIDFUMARAT; REG. NO/DATE: EU/1/15/1061 20151209 |
| 1301519 | PA2016009 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: TENOFOVIRO ALAFENAMIDAS; REGISTRATION NO/DATE: EU/1/15/1061/001 - 002 20151119 |
| 1663240 | 92854 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE RILPIVIRINE OU UNE FORME THERAPEUTIQUE EQUIVALENTE QUI EN DERIVE TELLE QUE PROTEGEE PAR LE BREVET DE BASE, TEL QU'UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE RILPIVIRINE, INCLUANT LE SEL CHLORHYDRATE DE RILPIVIRINE, ET TENOFOVIR, EN PARTICULIER LE FUMARATE DE TENOFOVIR DISOPROXIL |
| 0915894 | C00915894/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: TENOFOVIR DISOPROXILFUMARAT + EMTRICITABIN; REGISTRATION NUMBER/DATE: SWISSMEDIC 57316 21.03.2006 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


