VALSARTAN Drug Patent Profile
✉ Email this page to a colleague
When do Valsartan patents expire, and when can generic versions of Valsartan launch?
Valsartan is a drug marketed by Novitium Pharma, Alembic, Alkem Labs Ltd, Amneal Pharms, Aurobindo Pharma Ltd, Dr Reddys, Hetero Labs Ltd V, Ivax Pharms, Jubilant Generics, Lupin Ltd, Macleods Pharms Ltd, Mylan, Ohm Labs Inc, Prinston Inc, Sciegen Pharms Inc, Square Pharms, Torrent, Unichem, Watson Labs Inc, Apotex Inc, Mylan Pharms Inc, Watson Labs Teva, and Zydus Lifesciences. and is included in twenty-nine NDAs.
The generic ingredient in VALSARTAN is hydrochlorothiazide; valsartan. There are thirty-two drug master file entries for this compound. Twenty-one suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; valsartan profile page.
Summary for VALSARTAN
| US Patents: | 0 |
| Applicants: | 23 |
| NDAs: | 29 |
| Finished Product Suppliers / Packagers: | 30 |
| Raw Ingredient (Bulk) Api Vendors: | 152 |
| Clinical Trials: | 380 |
| Patent Applications: | 4,011 |
| Formulation / Manufacturing: | see details |
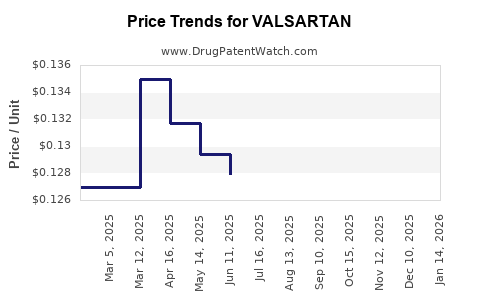
| Drug Prices: | Drug price information for VALSARTAN |
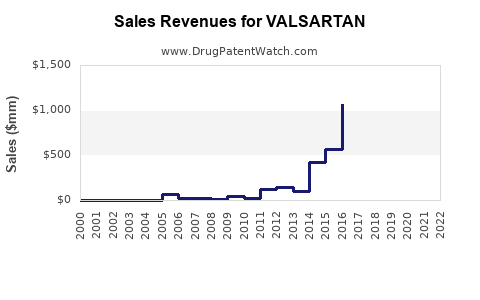
| Drug Sales Revenues: | Drug sales revenues for VALSARTAN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VALSARTAN |
| What excipients (inactive ingredients) are in VALSARTAN? | VALSARTAN excipients list |
| DailyMed Link: | VALSARTAN at DailyMed |
Recent Clinical Trials for VALSARTAN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Daewoong Pharmaceutical Co. LTD. | Phase 4 |
| CardioVascular Research Foundation, Korea | Phase 4 |
| Duk-Woo Park, MD | Phase 4 |
Pharmacology for VALSARTAN
| Drug Class | Angiotensin 2 Receptor Blocker |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists |


