Exelixis Company Profile
✉ Email this page to a colleague
What is the competitive landscape for EXELIXIS, and what generic alternatives to EXELIXIS drugs are available?
EXELIXIS has two approved drugs.
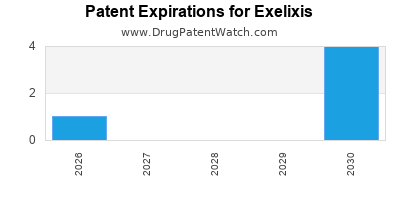
There are eleven US patents protecting EXELIXIS drugs.
There are two hundred and seven patent family members on EXELIXIS drugs in thirty-two countries and nineteen supplementary protection certificates in sixteen countries.
Drugs and US Patents for Exelixis
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-001 | Apr 25, 2016 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-001 | Apr 25, 2016 | RX | Yes | No | 11,098,015 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Exelixis | COMETRIQ | cabozantinib s-malate | CAPSULE;ORAL | 203756-002 | Nov 29, 2012 | RX | Yes | Yes | 11,091,440 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Exelixis Inc | CABOMETYX | cabozantinib s-malate | TABLET;ORAL | 208692-003 | Apr 25, 2016 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Exelixis Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Croatia | P20140563 | ⤷ Try a Trial |
| Eurasian Patent Organization | 201400110 | ⤷ Try a Trial |
| New Zealand | 779754 | ⤷ Try a Trial |
| Brazil | PI1006812 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Exelixis Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2213661 | PA2014033,C2213661 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: KABOZANTINIBAS IR JO FARMACINIU POZIURIU PRIIMTINOS DRUSKOS; REGISTRATION NO/DATE: EU/1/13/890/001 - EU/1/13/890/003 20140321 |
| 2213661 | C 2014 036 | Romania | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB SI ORICE FORMA ECHIVALENTA TERAPEUTIC AACESTUIA, INCLUSIV SARURILE ACCEPT DATE OF NATIONAL AUTHORISATION: 20140321; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/13/890/001, EU/1/13/890/002, EU/1/13/890/003; DATE OF FIRST AUTHORISATION IN EEA: 20140321 ABILE FARMACEUTIC; NATIONAL AUTHORISATION NUMBER: EU/1/13/890/001, EU/1/13/890/002, EU/1/13/890/003; |
| 2213661 | 220 50007-2014 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB; REGISTRATION NO/DATE: EU/1/13/890/001 - EU/1/13/890/003 20140326 |
| 2213661 | 43/2014 | Austria | ⤷ Try a Trial | PRODUCT NAME: CABOZANTINIB ODER EIN PHARMAZEUTISCH AKZEPTABLES SALZ DAVON; REGISTRATION NO/DATE: EU/1/13/890/001-003 (MITTEILUNG) 20140326 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.