PFIZER Company Profile
✉ Email this page to a colleague
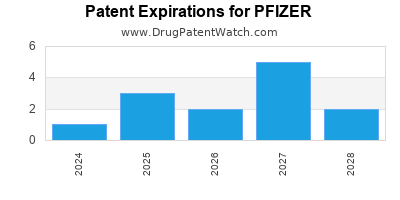
What is the competitive landscape for PFIZER, and when can generic versions of PFIZER drugs launch?
PFIZER has one hundred and ninety-five approved drugs.
There are forty-nine US patents protecting PFIZER drugs. There is one tentative approval on PFIZER drugs.
There are one thousand and twenty-seven patent family members on PFIZER drugs in seventy countries and two hundred and sixty-one supplementary protection certificates in nineteen countries.
Summary for PFIZER
| International Patents: | 1027 |
| US Patents: | 49 |
| Tradenames: | 159 |
| Ingredients: | 130 |
| NDAs: | 195 |
| Drug Master File Entries: | 3 |
| Patent Litigation for PFIZER: | See patent lawsuits for PFIZER |
| PTAB Cases with PFIZER as petitioner: | See PTAB cases with PFIZER as petitioner |
Drugs and US Patents for PFIZER
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | PFIZERPEN-AS | penicillin g procaine | INJECTABLE;INJECTION | 060286-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Pfizer | ZITHROMAX | azithromycin | FOR SUSPENSION;ORAL | 050693-001 | Sep 28, 1994 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | |||||
| Pfizer | TALZENNA | talazoparib tosylate | CAPSULE;ORAL | 217439-004 | Mar 7, 2024 | RX | Yes | No | 9,820,985 | ⤷ Sign Up | ⤷ Sign Up | ||||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-003 | Jan 14, 2022 | RX | Yes | Yes | 9,545,405 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Pfizer | TALZENNA | talazoparib tosylate | CAPSULE;ORAL | 211651-002 | Oct 16, 2018 | RX | Yes | Yes | 8,420,650 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Pfizer | MEDROL | methylprednisolone | TABLET;ORAL | 011153-001 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for PFIZER
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pfizer | XELJANZ XR | tofacitinib citrate | TABLET, EXTENDED RELEASE;ORAL | 208246-002 | Dec 12, 2019 | 6,965,027 | ⤷ Sign Up |
| Pfizer | TROVAN | trovafloxacin mesylate | TABLET;ORAL | 020759-001 | Dec 18, 1997 | 5,763,454 | ⤷ Sign Up |
| Pfizer | ZYVOX | linezolid | TABLET;ORAL | 021130-002 | Apr 18, 2000 | 5,688,792*PED | ⤷ Sign Up |
| Pfizer | DIFLUCAN IN DEXTROSE 5% IN PLASTIC CONTAINER | fluconazole | INJECTABLE;INJECTION | 019950-005 | Jul 8, 1994 | 4,404,216*PED | ⤷ Sign Up |
| Pfizer | FLAGYL ER | metronidazole | TABLET, EXTENDED RELEASE;ORAL | 020868-001 | Nov 26, 1997 | 6,103,262 | ⤷ Sign Up |
| Pfizer | TROVAN | trovafloxacin mesylate | TABLET;ORAL | 020759-002 | Dec 18, 1997 | 5,763,454 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for PFIZER drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | 5 mg and 10 mg | ➤ Subscribe | 2005-06-21 |
| ➤ Subscribe | For Injection | 500 mg/vial | ➤ Subscribe | 2011-06-17 |
| ➤ Subscribe | Oral Suspension | 100 mg/5 mL | ➤ Subscribe | 2009-08-03 |
| ➤ Subscribe | Injection | 2 mg/mL, 100 mL bag | ➤ Subscribe | 2009-12-29 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Delayed-release Tablets | 50 mg/0.2 mg | ➤ Subscribe | 2009-06-29 |
| ➤ Subscribe | Extended-release Tablets | 11 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Capsules | 20 mg, 40 mg, 60 mg and 80 mg | ➤ Subscribe | 2005-02-07 |
| ➤ Subscribe | Capsules | 75 mg, 100 mg and 125 mg | ➤ Subscribe | 2019-02-04 |
| ➤ Subscribe | Capsules | 0.125 mg, 0.25 mg, and 0.5 mg | ➤ Subscribe | 2014-05-01 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2005-12-21 |
| ➤ Subscribe | Injection | 2 mg/mL, 300 mL bag | ➤ Subscribe | 2009-09-01 |
| ➤ Subscribe | Tablets | 1 g | ➤ Subscribe | 2005-08-23 |
| ➤ Subscribe | Delayed-release Tablets | 75 mg/0.2 mg | ➤ Subscribe | 2008-11-28 |
| ➤ Subscribe | Extended-release Tablets | 4 mg and 8 mg | ➤ Subscribe | 2012-10-31 |
| ➤ Subscribe | Injection | 20 mg/mL, 2 mL and 5 mL vials | ➤ Subscribe | 2004-07-26 |
International Patents for PFIZER Drugs
Supplementary Protection Certificates for PFIZER Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2488512 | C202230042 | Spain | ⤷ Sign Up | PRODUCT NAME: RIMEGEPANT O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/22/1645; DATE OF AUTHORISATION: 20220425; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/22/1645; DATE OF FIRST AUTHORISATION IN EEA: 20220425 |
| 2767537 | 132019000000153 | Italy | ⤷ Sign Up | PRODUCT NAME: TALAZOPARIB(TALZENNA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/19/1377, 20190624 |
| 1666481 | 37/2017 | Austria | ⤷ Sign Up | PRODUCT NAME: TOFACITINIB, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH VERTRAEGLICHEN SALZES, EINSCHLIESSLICH DES CITRATS; REGISTRATION NO/DATE: EU/1/17/1178 (MITTEILUNG) 20170324 |
| 1470124 | C 2017 016 | Romania | ⤷ Sign Up | PRODUCT NAME: PALBOCICLIB OPTIONAL SUB FORMA DE SARE, ESTER, AMIDA SAUPROMEDICAMENT AL ACESTUIA, ACCEPTABILEFARMACEUTIC; NATIONAL AUTHORISATION NUMBER: EU/1/16/1147; DATE OF NATIONAL AUTHORISATION: 20161109; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/16/1147; DATE OF FIRST AUTHORISATION IN EEA: 20161109 |
| 0266730 | 98C0014 | Belgium | ⤷ Sign Up | PRODUCT NAME: MIGLITOL; NAT. REGISTRATION NO/DATE: NL 22 138 19970204; FIRST REGISTRATION: NL - 19 343 UR 19960723 |
| 0717738 | SPC/GB01/025 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: LINEZOLID; REGISTERED: UK PL 00032/0259 20010105; UK PL 00032/0260 20010105; UK PL 00032/0261 20010105; UK PL 00032/0262 20010105 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.