Abbvie Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ABBVIE, and when can generic versions of ABBVIE drugs launch?
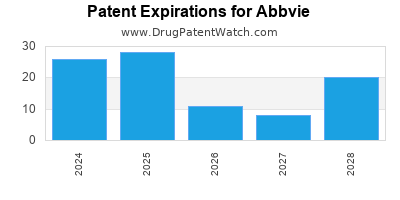
ABBVIE has one hundred and thirty-eight approved drugs.
There are two hundred and twenty-nine US patents protecting ABBVIE drugs.
There are two thousand seven hundred and twenty-eight patent family members on ABBVIE drugs in sixty-six countries and three hundred and ninety-five supplementary protection certificates in nineteen countries.
Summary for Abbvie
| International Patents: | 2728 |
| US Patents: | 229 |
| Tradenames: | 124 |
| Ingredients: | 99 |
| NDAs: | 138 |
| Drug Master File Entries: | 1 |
| Patent Litigation for Abbvie: | See patent lawsuits for Abbvie |
Drugs and US Patents for Abbvie
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbvie | VIEKIRA PAK (COPACKAGED) | dasabuvir sodium; ombitasvir, paritaprevir, ritonavir | TABLET;ORAL | 206619-001 | Dec 19, 2014 | DISCN | Yes | No | 8,501,238 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Abbvie | NIMBEX PRESERVATIVE FREE | cisatracurium besylate | INJECTABLE;INJECTION | 020551-002 | Dec 15, 1995 | AP | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ||||
| Abbvie | FML | fluorometholone | SUSPENSION/DROPS;OPHTHALMIC | 016851-002 | Jul 28, 1982 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ||||
| Abbvie | VIBERZI | eluxadoline | TABLET;ORAL | 206940-001 | May 27, 2015 | RX | Yes | No | 9,364,489 | ⤷ Sign Up | ⤷ Sign Up | ||||
| Abbvie | MAVYRET | glecaprevir; pibrentasvir | PELLETS;ORAL | 215110-001 | Jun 10, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | |||||
| Abbvie | UBRELVY | ubrogepant | TABLET;ORAL | 211765-002 | Dec 23, 2019 | RX | Yes | Yes | 8,912,210 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Abbvie | QULIPTA | atogepant | TABLET;ORAL | 215206-002 | Sep 28, 2021 | RX | Yes | No | 10,117,836 | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Abbvie
Paragraph IV (Patent) Challenges for ABBVIE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Opthalmic Solution | 0.45% | ➤ Subscribe | 2011-08-23 |
| ➤ Subscribe | Extended-release Tablets | 500 mg | ➤ Subscribe | 2005-02-08 |
| ➤ Subscribe | Capsules | 7 mg/10 mg | ➤ Subscribe | 2016-09-26 |
| ➤ Subscribe | Ophthalmic Solution | 0.10% | ➤ Subscribe | 2006-12-20 |
| ➤ Subscribe | Extended-release Tablets | 2 mg/180 mg and 2 mg/240 mg | ➤ Subscribe | 2007-11-09 |
| ➤ Subscribe | Oral Solution | 80 mg/20 mg per mL | ➤ Subscribe | 2014-06-19 |
| ➤ Subscribe | Ophthalmic Solution | 0.2%/0.5% | ➤ Subscribe | 2008-11-21 |
| ➤ Subscribe | Extended-release Tablets | 4 mg/ 240 mg | ➤ Subscribe | 2007-07-24 |
| ➤ Subscribe | Tablets | 100 mg | ➤ Subscribe | 2010-12-21 |
| ➤ Subscribe | Ophthalmic Solution | 0.03% | ➤ Subscribe | 2008-12-22 |
| ➤ Subscribe | Tablets | 1 mg, 2 mg and 4 mg | ➤ Subscribe | 2004-10-04 |
| ➤ Subscribe | Injection | 0.002 mg per mL in 1 mL vial and 0.005 mg per mL in 1 mL and 2 mL vials | ➤ Subscribe | 2008-11-28 |
| ➤ Subscribe | Ophthalmic Solution | 0.5% | ➤ Subscribe | 2010-12-07 |
| ➤ Subscribe | Delayed-release Capsules | 45 mg | ➤ Subscribe | 2009-09-02 |
| ➤ Subscribe | Tablets | 12.5 mg, 25 mg, 50 mg, and 100 mg | ➤ Subscribe | 2013-01-14 |
| ➤ Subscribe | Capsules | 4 mcg | ➤ Subscribe | 2008-08-25 |
| ➤ Subscribe | Capsules | 5 mg | ➤ Subscribe | 2005-08-17 |
| ➤ Subscribe | Extended-release Capsules | 20 mg, 40 mg, 80 mg and 120 mg | ➤ Subscribe | 2017-07-25 |
| ➤ Subscribe | Tablets | 48 mg | ➤ Subscribe | 2008-07-01 |
| ➤ Subscribe | Gel | 10% | ➤ Subscribe | 2014-06-19 |
| ➤ Subscribe | Injection | 10 mg/mL (2 mL) | ➤ Subscribe | 2018-07-13 |
| ➤ Subscribe | Extended-release Capsules | 28 mg | ➤ Subscribe | 2013-06-12 |
| ➤ Subscribe | Tablets | 2.5 mg/200 mg | ➤ Subscribe | 2006-02-24 |
| ➤ Subscribe | Injection | 2 mg/mL, 10 mL vial | ➤ Subscribe | 2009-08-12 |
| ➤ Subscribe | Delayed-release Tablets | 800 mg | ➤ Subscribe | 2011-07-13 |
| ➤ Subscribe | Extended-release Capsules | 7 mg, 14 mg, 21 mg, and 28 mg | ➤ Subscribe | 2013-06-10 |
| ➤ Subscribe | Tablets | 10 mg, 20 mg, and 40 mg | ➤ Subscribe | 2015-01-21 |
| ➤ Subscribe | Ophthalmic Solution | 0.40% | ➤ Subscribe | 2005-01-28 |
| ➤ Subscribe | Extended-release Tablets | 250 mg | ➤ Subscribe | 2004-05-03 |
| ➤ Subscribe | Capsules | 21 mg/10 mg | ➤ Subscribe | 2016-09-23 |
| ➤ Subscribe | Opthalmic Solution | 0.45% | ➤ Subscribe | 2011-08-24 |
| ➤ Subscribe | Extended-release Tablets | 7.5 mg and 15 mg | ➤ Subscribe | 2008-12-22 |
| ➤ Subscribe | Tablets | 100 mg/25 mg | ➤ Subscribe | 2008-12-23 |
| ➤ Subscribe | Ophthalmic Solution | 0.15% | ➤ Subscribe | 2006-11-03 |
| ➤ Subscribe | Extended-release Tablets | 1 mg/240 mg | ➤ Subscribe | 2008-02-20 |
| ➤ Subscribe | Capsules | 145 mcg and 290 mcg | ➤ Subscribe | 2016-08-30 |
| ➤ Subscribe | Ophthalmic Solution | 0.25% | ➤ Subscribe | 2014-07-30 |
| ➤ Subscribe | Tablets | 5 mg/80 mg | ➤ Subscribe | 2017-06-09 |
| ➤ Subscribe | Capsules | 100 mg | ➤ Subscribe | 2012-10-31 |
| ➤ Subscribe | Ophthalmic Solution | 0.01% | ➤ Subscribe | 2011-04-05 |
| ➤ Subscribe | Delayed-release Capsules | 400 mg | ➤ Subscribe | 2014-06-17 |
| ➤ Subscribe | Tablets | 5 mg and 10 mg | ➤ Subscribe | 2007-10-16 |
| ➤ Subscribe | Capsules | 1 mcg and 2 mcg | ➤ Subscribe | 2008-10-14 |
| ➤ Subscribe | Topical Solution | 0.03% | ➤ Subscribe | 2010-05-03 |
| ➤ Subscribe | Delayed-release Capsules | 135 mg | ➤ Subscribe | 2009-09-01 |
| ➤ Subscribe | Tablets | 400 mg and 600 mg | ➤ Subscribe | 2010-05-10 |
| ➤ Subscribe | Gel | 7.5% | ➤ Subscribe | 2017-02-13 |
| ➤ Subscribe | Capsules | 10 mg and 20 mg | ➤ Subscribe | 2005-03-30 |
| ➤ Subscribe | Extended-release Capsules | 7 mg, 14 mg, 21 mg, and 28 mg | ➤ Subscribe | 2013-08-16 |
| ➤ Subscribe | Tablets | 145 mg | ➤ Subscribe | 2007-10-19 |
| ➤ Subscribe | Injection | 2 mg/mL, 5 mL vial and 10 mg/mL, 20 mL vial | ➤ Subscribe | 2009-08-04 |
| ➤ Subscribe | Suppository | 1000 mg | ➤ Subscribe | 2013-05-24 |
| ➤ Subscribe | Extended-release Capsules | 7 mg, 14 mg, 21 mg | ➤ Subscribe | 2013-06-17 |
| ➤ Subscribe | Tablets | 5 mg/200 mg | ➤ Subscribe | 2005-05-27 |
| ➤ Subscribe | Injection | 400 mg/vial and 600 mg/vial | ➤ Subscribe | 2014-10-29 |
| ➤ Subscribe | Extended-release Tablet | 500 mg | ➤ Subscribe | 2010-12-06 |
| ➤ Subscribe | Capsules | 14 mg/10 mg and 28 mg/10 mg | ➤ Subscribe | 2015-05-18 |
International Patents for Abbvie Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Portugal | 1725537 | ⤷ Sign Up |
| Israel | 178040 | ⤷ Sign Up |
| Lithuania | 3213750 | ⤷ Sign Up |
| Hong Kong | 1243761 | ⤷ Sign Up |
| China | 103328478 | ⤷ Sign Up |
| Japan | 5367946 | ⤷ Sign Up |
| World Intellectual Property Organization (WIPO) | 2006130174 | ⤷ Sign Up |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Abbvie Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1534313 | 2015/071 | Ireland | ⤷ Sign Up | PRODUCT NAME: AN OCULAR IRRIGATION SOLUTION COMPRISING PHENYLEPHRINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF AND KETOROLAC OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/15/1018 20150728 |
| 2176234 | SPC/GB17/019 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: ELUXADOLINE; REGISTERED: UK EU/1/16/1126/001(NI) 20160921; UK EU/1/16/1126/002(NI) 20160921; UK EU/1/16/1126/003(NI) 20160921; UK EU/1/16/1126/004(NI) 20160921; UK PLGB 45496/0015 20160921 |
| 2368890 | 300731 | Netherlands | ⤷ Sign Up | PRODUCT NAME: OMBITASVIR OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/14/982 20150119 |
| 2618831 | 17C1039 | France | ⤷ Sign Up | PRODUCT NAME: GLECAPREVIR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE OU UN ESTER DE CELUI-CI; REGISTRATION NO/DATE: EU/1/17/1213 20170728 |
| 0502314 | SPC/GB02/037 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND HYDROCHLOROTHIAZIDE; REGISTERED: UK EU/1/02/213/001 20020419; UK EU/1/02/213/002 20020419; UK EU/1/02/213/003 20020419; UK EU/1/02/214/004 20020419; UK EU/1/02/213/005 20020419; UK EU/1/02/213/006 20020419; UK EU/1/02/213/007 20020419; UK EU/1/02/213/008 20020419; UK EU/1/02/213/009 20020419; UK EU/1/02/213/010 20020419 |
| 0334429 | 97C0002 | Belgium | ⤷ Sign Up | PRODUCT NAME: ESTRADIOL; NAT. REGISTRATION NO/DATE: NL 18978 19960731; FIRST REGISTRATION: SE - 11783 19930305 |
| 2435432 | 386 50008-2017 | Slovakia | ⤷ Sign Up | PRODUCT NAME: VENETOKLAX; REGISTRATION NO/DATE: EU/1/16/1138 20161207 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.